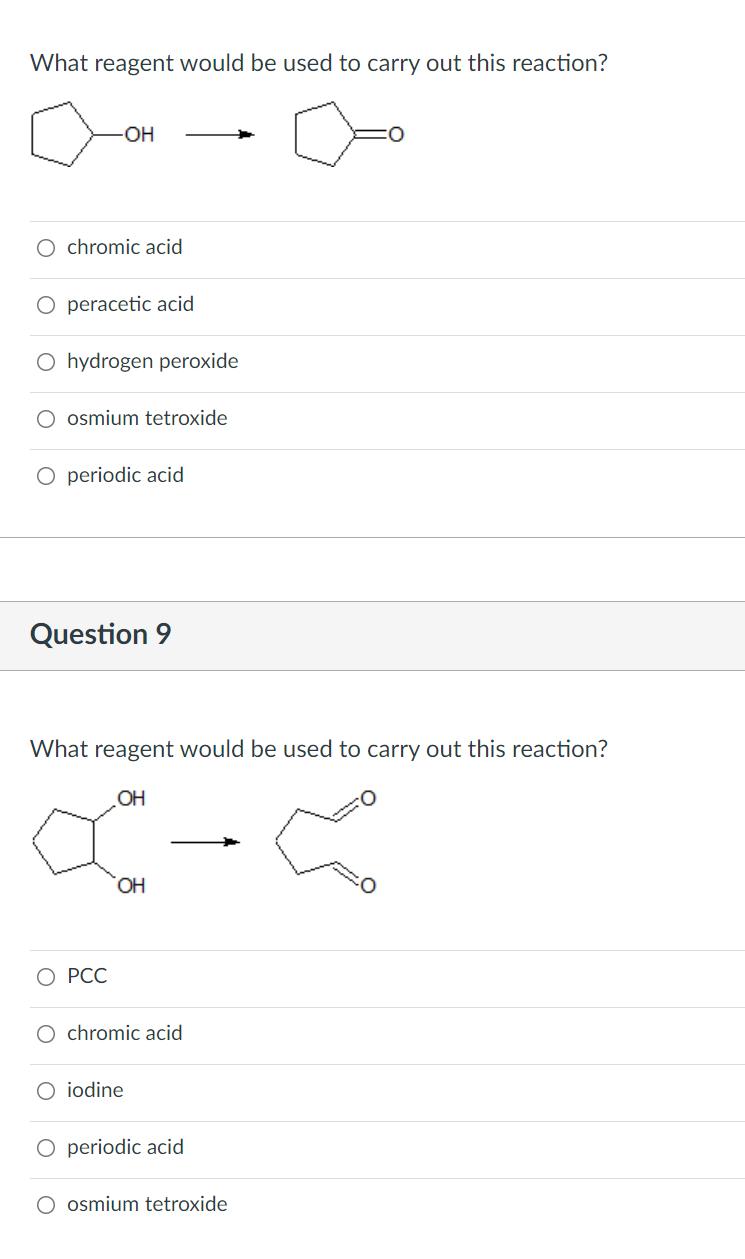
What reagent would be used to carry out this reaction? OH O chromic acid peracetic acid...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
What reagent would be used to carry out this reaction? OH O chromic acid peracetic acid O hydrogen peroxide O osmium tetroxide O periodic acid Question 9 What reagent would be used to carry out this reaction? OH HO. О РСС chromic acid O iodine O periodic acid O osmium tetroxide What reagent would be used to carry out this reaction? OH O chromic acid peracetic acid O hydrogen peroxide O osmium tetroxide O periodic acid Question 9 What reagent would be used to carry out this reaction? OH HO. О РСС chromic acid O iodine O periodic acid O osmium tetroxide
Expert Answer:
Answer rating: 100% (QA)
The reagent is carried out the oxidation of secondary alcohol into the ketone is the chromic acid Th... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
What reagents could be used to carry out the following syntheses? RCH2CH RCH-CH2 RCH2CH2Br RCHCH3 Br Br RCCH Br RCCH3 RC-CH Br RCH2CH
-
What reagent would be used to carry out this reaction? HS. SH O osmium tetroxide O periodic acid O chromic acid O iodine
-
What process do you think would be used to analyze the simulation results?
-
A money market portfolio has a market value of $20,000,000 and its value will change by $500 for a change in short-term yields of one basis point. The eurodollar futures contract has a tick size of...
-
During its preliminary review of the financial statements of Barton, Inc., Simon and Associates, CPA discovered a lack of proper segregation of duties between the programming and operating functions...
-
1. Stella Scott has a work-study position at the fitness center on campus. Her manager has asked her to create a worksheet to keep track of attendance and revenue for the fitness center's classes....
-
A study of 3,005 adults ages 57 to 85 showed that 82% of them use at least one prescription drug. The margin of error is 2 percentage points (based on data from Use of Prescription and...
-
Multiple Choice. Choose the best answer. 1. Special purpose governments differ from general purpose governments in that special purpose governments: a. Provide a single function or limited range of...
-
Aria Kim is a Marketing Director for Petopia Products, a manufacturer of pet products with an extensive product catalogue and multiple product lines. With her recent promotion, she is responsible for...
-
Reconsider the Lockhead Aircraft Co. problem presented in Problem 16.15 regarding a project to develop a new fighter airplane for the U.S. Air Force. Management is extremely concerned that current...
-
On January 1, 2019, a company reported assets of $1900000 and liabilities of $600000. During 2019, assets decreased by $100,000 and Stockholder's Equity decreased $200,000. What is the amount of...
-
The increasing cost of college has reduced how much young adults have saved for retirement. How does a higher cost of college change the life-cycle pattern of saving?
-
One analyst predicts that self-driving cars will ultimately reduce the number of cars that are produced. She argues that because self-driving cars can drive other people rather than sitting in...
-
How can the Federal Reserve influence long-term interest rates, and shift the MP curve, without changing the current risk-free interest rate?
-
An example of U.K. foreign investment is the aerospace manufacturing plant in Austin, Texas, that is owned by the U.K.-owned BAE Systems. Describe how this investment affects the three inputs into...
-
After reading a report that says that around 70% of U.S. GDP is consumption, your friend Alex states, Spending 70% of GDP on consumption is a lot. All people care about is buying stuff, consuming...
-
When does a buyer need to tell the seller that the buyer will be using an FHA-insured loan to purchase the property? OA) Never-the seller doesn't care OB) In the buyer's initial offer OC) After the...
-
Based on the scenario described below, generate all possible association rules with values for confidence, support (for dependent), and lift. Submit your solutions in a Word document (name it...
-
The rate constant for a reaction can be increased by _____ the stability of the reactant or by _____ the stability of the transition state.
-
Bupropion hydrochloride is an antidepressant marketed under the trade name Wellbutrin®. Propose a synthesis of bupropion hydrochloride, starting with benzene. Cl CCHCH3 NH2C(CH3)3CI bupropion...
-
What alkene would you treat with a peroxyacid in order to obtain each of the following epoxides? a. b. c. d. H2C CHCH2CH3 H3C H3C CH2CH3
-
Two quality managers meet for lunch every Friday to discuss common problems, compare notes, and make suggestions to each other. Today their discussion has turned into a debate. The topic is the...
-
The Office of Technology Assessment (OTA) reports that only 11 to 15 percent of all machine tools in the U.S. are automated; the majority of those automated are found in large companies. A Census...
-
Pennsylvanias Department of Community and Economic Development maintains a network of industrial resource centers (IRCs) that are operated as nonprofit corporations managed by the private sector but...

Study smarter with the SolutionInn App


