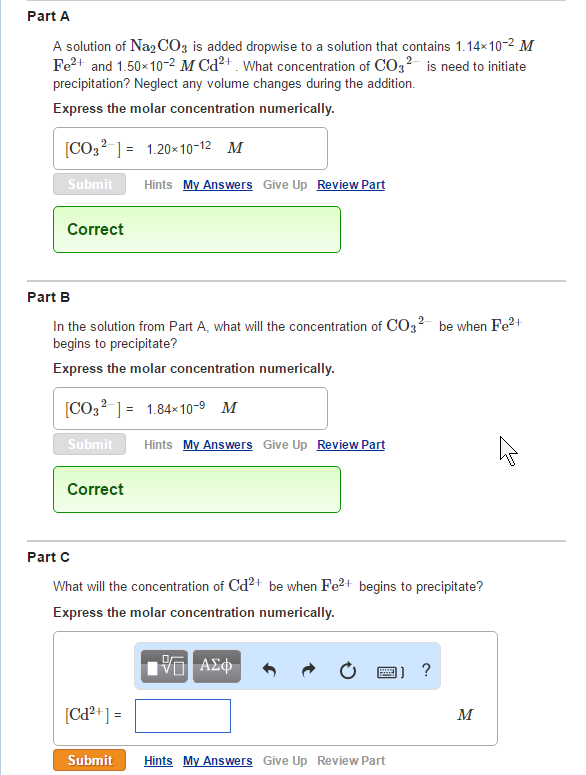
What will the concentration of Cd2+ be when Fe2+ begins toprecipitate? Part A A solution of Na2CO3
Fantastic news! We've Found the answer you've been seeking!
Question:
What will the concentration of Cd2+ be when Fe2+ begins toprecipitate?
Transcribed Image Text:
Part A A solution of Na2CO3 is added dropwise to a solution that contains 1.14×10-² M Fe²+ and 1.50×10-2 M Cd²+ What concentration of CO2 is need to initiate precipitation? Neglect any volume changes during the addition. Express the molar concentration numerically. [CO3²] 1.20x10-12 M Submit Correct Part B In the solution from Part A, what will the concentration of CO32- be when Fe²+ begins to precipitate? Express the molar concentration numerically. Hints My Answers Give Up Review Part [CO3²] = 1.84x10-⁹ M Submit Correct [Cd²+] = Hints My Answers Give Up Review Part Part C What will the concentration of Cd²+ be when Fe²+ begins to precipitate? Express the molar concentration numerically. ΑΣΦ Submit Hints My Answers Give Up Review Part M Part A A solution of Na2CO3 is added dropwise to a solution that contains 1.14×10-² M Fe²+ and 1.50×10-2 M Cd²+ What concentration of CO2 is need to initiate precipitation? Neglect any volume changes during the addition. Express the molar concentration numerically. [CO3²] 1.20x10-12 M Submit Correct Part B In the solution from Part A, what will the concentration of CO32- be when Fe²+ begins to precipitate? Express the molar concentration numerically. Hints My Answers Give Up Review Part [CO3²] = 1.84x10-⁹ M Submit Correct [Cd²+] = Hints My Answers Give Up Review Part Part C What will the concentration of Cd²+ be when Fe²+ begins to precipitate? Express the molar concentration numerically. ΑΣΦ Submit Hints My Answers Give Up Review Part M
Expert Answer:
Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
What is the I concentration just as AgCl begins to precipitate when 1.0 M AgNO3 is slowly added to a solution containing 0.015 M Cl and 0.015 M I?
-
What is the Cl concentration just as Ag2CrO4 begins to precipitate when 1.0 MAgNO3 is slowly added to a solution containing 0.015 M Cl and 0.015 M CrO42?
-
When methyl iodide at 0.I M concentration is allowed to react with sodium ethoxide at 0.1 M concentration in ethanol solution, the product ethyl methyl ether is obtained in good yield. Explain why...
-
Panamint Candy Company prepared the following amortization table for $300,000 of 5-year, 9% bonds issued and sold by Panamint on January 1, 2021, for $285,000: Required: 1. Prepare the entry to...
-
What are convertible bonds? How do they benefit both the investor and the issuer?
-
Use the following data (in millions) for Dell, Inc., for a recent year to answer the questions below: Retained earnings, beginning of year ....... $24,744 Retained earnings, end of year ...........
-
In spring 1989, Michael Jordan and the Chicago Bulls were in Indianapolis, Indiana, to play against the Indiana Pacers. At the same time, Karla Knafel was singing with a band at a hotel in...
-
Westside Auto purchases a component used in the manufacture of automobile generators directly from the supplier. Westsides generator production operation, which is operated at a constant rate, will...
-
3. (a) Find the binary representation of /3. (b) By noting that -9.6= (-1) 23 1.2. x Use the result in (a), represent -9.6 in IEEE single precision format through rounding. (c) Represent the result...
-
In the current year (year 0), Amisha became a shareholder in Sultan Inc., a calendar year S corporation, by contributing $15,000 cash in exchange for stock. Shortly before the end of the year,...
-
Solve the following inequality and graph the solution: (x 5)(x + 5) > 0 Choose test values and indicate whether the inequality is true or false in each region. Enter the test values from smallest to...
-
Looking for help with the Minimum and Maximum part of this code... .js file with the following functions: average, minimum, maximum. All functions will take an array as an input and the calculated...
-
Part 2: Complete the following calculations Quantity Price Total Revenue Marginal Revenue 0 $22 1 $20 2 $18 3 $16 4 $14 5 $12 6 $10 7 $8 8 $6
-
function name (price, qty, taxRate) a. Three numbers (decimal values) are passed to this function. First parameter holds the unit price of an item. Second parameter holds the quantity ordered and...
-
File/directory processing Write a Java, C# or C/C++ program (the choice is yours) for file/directory processing according to the following rules. The program requested for this project must have a...
-
Find and simplify (h = 0) f(a + h) f(a) h for the following function. f(x) = 8x - 4x + 1 X
-
Hawaii Surf shop has a retail store on Oahu selling surfboards and other surf apparel. Their GET license is GE 123-111-5523-01. And the last 4 digits of their EIN is 8825 They had the following sales...
-
One of the significant and relevant accounts for this cycle is equipment. For this account, what would typically be the most relevant assertions for the auditor to consider? Why is it important for...
-
The electrical conductivity of titanium is approximately 2500 times greater than that of silicon. Titanium has a hexagonal close-packed structure, and silicon has the diamond structure. Explain how...
-
Does the reactivity of a metal correlate with its first ionization energy? Explain.
-
The semiconductor GaP has a band gap of 2.2 eV. Green LEDs are made from pure GaP. What wavelength of light would be emitted from an LED made from GaP?
-
What is the adverse selection problem?
-
Automobile insurance companies charge lower rates to married individuals than they do to unmarried individuals. What economic reason is there for such a practice? Is it fair?
-
An advanced degree is required to teach at most colleges. In what sense is this a form of restricting entry through licensure?

Study smarter with the SolutionInn App



