When 15.0 mL of a 1.53x104 M nickel(II) acetate solution is combined with 18.0 mL of...
Fantastic news! We've Found the answer you've been seeking!
Question:
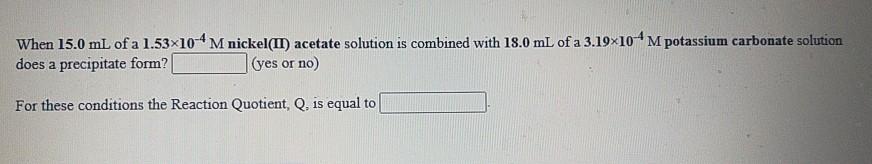
Transcribed Image Text:
When 15.0 mL of a 1.53x104 M nickel(II) acetate solution is combined with 18.0 mL of a 3.19×10M potassium carbonate solution does a precipitate form? (yes or no) For these conditions the Reaction Quotient, Q, is equal to When 15.0 mL of a 1.53x104 M nickel(II) acetate solution is combined with 18.0 mL of a 3.19×10M potassium carbonate solution does a precipitate form? (yes or no) For these conditions the Reaction Quotient, Q, is equal to
Expert Answer:
Answer rating: 100% (QA)
Initial condition is Q Ksp so that precipitate would be form Calculation ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
if you dilute 100 ml of a 1 mM HCL solution with 400 ml of water, what would you predict the new PH to be
-
Solution A was prepared by dissolving potassium acetate in methanol. Solution B was prepared by adding potassium methoxide to acetic acid. Reaction of methyl iodide either with solution A or with...
-
Potassium sulfate solution reacts with barium bromide solution to produce a precipitate of barium sulfate and a solution of potassium bromide. Write the molecular equation for this reaction. Then...
-
In recent years, Avery Transportation purchased three used buses. Because of frequent turnover in the accounting department, a different accountant was in charge of selecting the depreciation method...
-
Refer to the Excel display below and answer the given questions. Construct the multiple regression equation that expresses earthquake magnitude in terms of latitude, longitude, and depth.
-
In the last column for NYTD,figure out how NYT'sDNA elements will impact their performance. What DNA elements should NYTD keep, what should it jettison, and what should it change? A element...
-
Vibration spectra can have many frequency peaks. What is key to simplifying the analysis of the number of peaks of interest?
-
Altuve Co. was incorporated on January 1, 2013, at which time 500,000 shares of $1 par value common stock were authorized, and 210,000 of these shares were issued for $15 per share. Net income for...
-
Write a javascript program to display the grade of a student 1) Text 1-Student Name 2) Text 2-Student Id 3) Text 3 - Mark 4) Button - When you click the button display the grade in Paragraph (apply...
-
A receiver for base band digital data has a threshold set at instead of zero. Rederive (9.8), (9.9), and (9.11) taking this into account. If P(+A) = P(-A) = 1/2, find E b / N 0 in decibels as a...
-
Brady leaves her diamond necklace to be repaired at Diamonds R Us, a jewelry store that sells new and used diamond jewelry, Diamonds R Us sells the necklace to Doris, who does not know that it...
-
How does the process of reverse innovation work?
-
What four questions could be used to organize the presentation of a real-time balanced scorecard in the executive dashboard for a small business?
-
An organization that takes the lead in addressing emerging social issues is being ____________, showing the most progressive corporate social responsibility strategy. (a) accommodative (b) defensive...
-
Advice to study a job, carefully train workers to do that job, and link financial incentives to job performance would most likely come from ____________. (a) scientific management (b) contingency...
-
With respect to return on assets (ROA) and the debt ratio, the preferred directions when analyzing them from a control standpoint are ________. (a) decrease ROA, increase debt (b) increase ROA,...
-
How Globalization has affected unemployment in America ? Your own response detailing what you learned about the topic after researching it. For example were your original ideas about the topic...
-
On January 1, 2018, Khalid Ltd., which follows IAS 17, entered into an eight-year lease agreement for three dryers. Annual lease payments for the equipment are $28,500 at the beginning of each lease...
-
Consider structural formulas A, B, C, and D: (a) Which structures contain a positively charged carbon? (b) Which structures contain a positively charged nitrogen? (c) Which structures contain a...
-
Represent the reaction of chlorine with each of the enol forms of 2-butanone according to the curved arrow formalism just described. For Information: (see Problem 18.3)
-
From among the carbohydrates shown in Problem 25.20 choose the one(s) that (a) Belong to the L series (b) Are deoxy sugars (c) Are branched-chain sugars (d) Are ketoses (e) Are furanose forms (f)...
-
Determine the longitudinal tensile strength of the hybrid carbon/aramid/ epoxy composite described in Problem 3.4 and Figure 3.10 of Chapter 3 if the fiber packing array is square with the closest...
-
For the IM-9/8551-7 carbon/epoxy composite rod design of Problem 3.3 in Chapter 3, what would be the increase in the longitudinal tensile strength compared with that of the original 6061-T6 aluminum...
-
Assuming that the failure mode for longitudinal compression of unidirectional E-glass/epoxy with fiber volume fraction \(v_{\mathrm{f}}=0.6\) is a transverse tensile rupture due to Poisson strains,...

Study smarter with the SolutionInn App


