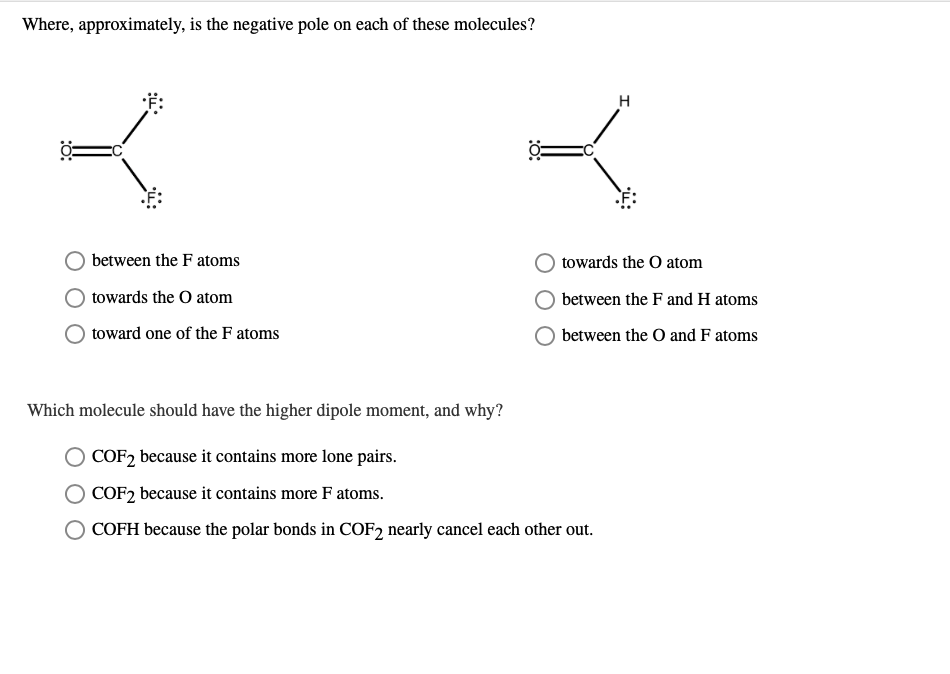
Where, approximately, is the negative pole on each of these molecules? between the F atoms towards...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Where, approximately, is the negative pole on each of these molecules? between the F atoms towards the O atom toward one of the F atoms H towards the O atom between the F and H atoms between the O and F atoms Which molecule should have the higher dipole moment, and why? COF2 because it contains more lone pairs. COF2 because it contains more F atoms. COFH because the polar bonds in COF2 nearly cancel each other out. Where, approximately, is the negative pole on each of these molecules? between the F atoms towards the O atom toward one of the F atoms H towards the O atom between the F and H atoms between the O and F atoms Which molecule should have the higher dipole moment, and why? COF2 because it contains more lone pairs. COF2 because it contains more F atoms. COFH because the polar bonds in COF2 nearly cancel each other out.
Expert Answer:
Answer rating: 100% (QA)
Answer 1 dipole moment due to two F will point opposite direction of ... View the full answer

Related Book For 

Conceptual Physical Science
ISBN: 978-0134060491
6th edition
Authors: Paul G. Hewitt, John A. Suchocki, Leslie A. Hewitt
Posted Date:
Students also viewed these chemistry questions
-
Determine whether each of these molecules is chiral, for those that are chiral, put an asterisk at the chirality center. b) a) d) I e) )
-
Perform the Gram-Schmidt process on each of these bases for R2. (a) (b) (c) Then turn those orthogonal bases into orthonormal bases. G) G) 1 0)(3)
-
Perform the Gram-Schmidt process on each of these bases for R3. (a) (b) Then turn those orthogonal bases into orthonormal bases. 10 600 01/
-
Your geothermal heat pump is used to heat your house in the winter. The heat pump is operated as a vapor-compression cycle. The design is such that there must be a 10 degree temperature difference...
-
Brick Oven Corporation was organized early in 2015. The following expenditures were made during the first few months of the year: Attorneys fees to organize the corporation ..... $ 9,000 Purchase of...
-
Pan American Refineries, headquartered in Houston, must decide among three sites for the construction of a new oil-processing center. The firm has selected the six factors listed below as a basis for...
-
Repeat Example 7.3 using \(10 \mathrm{~kg} / \mathrm{h}\) of solvent in each stage. Data From Example 7.3:- The feed of Example 7.2 is extracted three times with pure chloroform at 298 K, using 8...
-
Dorsey Company manufactures three products from a common input in a joint processing operation. Joint processing costs up to the split-off point total $350,000 per quarter. The company allocates...
-
Exercise 1 (8 points). Prove that, for all n = N, (Hint:) you may try by induction. 5|(n-n). Exercise 2 (9 points). Solve the following systems of congruences. (a) (b) x = 6 (mod 10) x = 11 (mod 15)...
-
Lois is a full-time delivery driver for Frank's Frozen Foods, and she gets reimbursed $0.46 for each mile driven, plus $310 a month for wear and tear on her car. She has just received a job offer...
-
Minimize and maximize f(x)=-X? -0.5 x2 subjected to 2x1 +3x2 -1220, X1 +X2 -8 <0, 1/x1-0.5x2 <0 - Xi <0, - X2 <0. Plot the feasible region (use MATLAB) and find the x* and f(x*) at the minimum and...
-
Family Law 1- What is "the presumption of resulting trust"? 2- What is a "constructive trust"? 3- What is a "capital gain"? 4- What is the "adjusted cost base" of a property? 5- What is the "adjusted...
-
What are the long-term implications of social change for systems of governance, community resilience, and collective well-being, and how can participatory approaches to social innovation foster...
-
With reference to decided cases and other authorities, compare and contrast how the following unions are dealt with in the Namibian and South African legal systems: 1.1 Civil Unions 1.2 Co-Habitation...
-
A piece of newly purchased industrial equipment costs $728143 and is classified as seven-year property under MACRS. What is the book value at the beginning of year 8? (Round your final answer to the...
-
You find the following order book on a particular stock. The last trade on the stock was at $58.34. Buy Orders Sell Orders Shares Price Shares Price 250 $58.33 250 $58.36 200 58.32 800 58.37 900 175...
-
The following are the accounts of United Forward Trading Limited, a company that engages in Shipping and container services for the years ended 31 December 2014 and 215 respectively. Statements of...
-
D Which of the following is considered part of the Controlling activity of managerial accounting? O Choosing to purchase raw materials from one supplier versus another O Choosing the allocation base...
-
Distinguish between dc and ac.
-
Two cars are raised to the same elevation on service station lifts. If one car is twice as massive as the other, how do their potential energies compare?
-
What are some examples of alkaloids?
-
There is another possible explanation for purchased goodwill appearing in a sole proprietor's statement of financial position. What do you think it might be?
-
Why do the assets need to be revalued in these cases? The business has not been sold.
-
The shown partners have always shared profits and losses in the ratio: Holt 4; Stott 2: Young 1. From 1 January the assets were to be revalued as the profit sharing ratios are to be altered soon. The...

Study smarter with the SolutionInn App


