Which of the following are desired properties of the aggregate production function? (Make sure you understand...
Fantastic news! We've Found the answer you've been seeking!
Question:
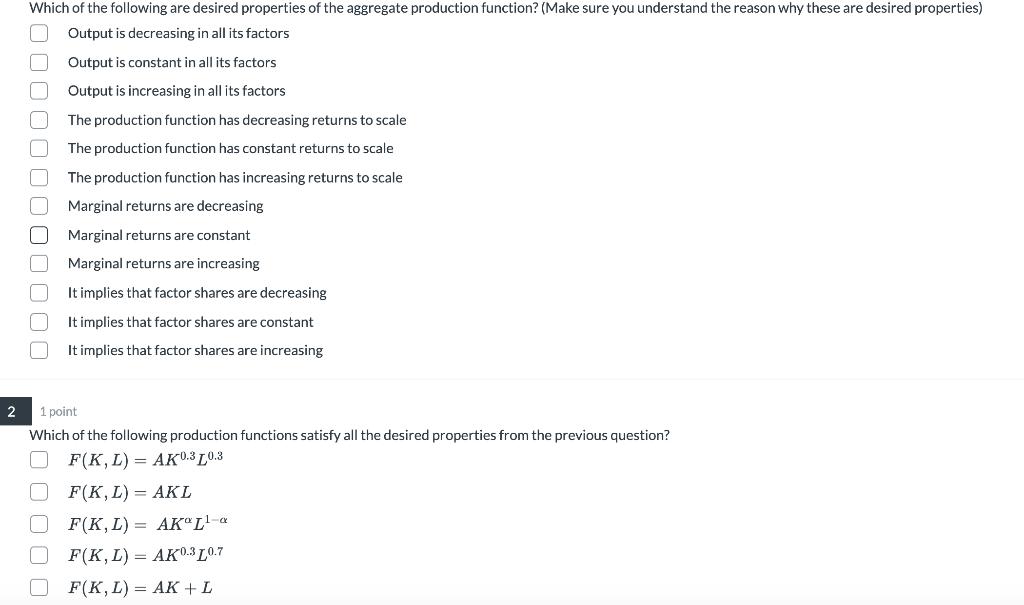
Transcribed Image Text:
Which of the following are desired properties of the aggregate production function? (Make sure you understand the reason why these are desired properties) Output is decreasing in all its factors Output is constant in all its factors Output is increasing in all its factors. The production function has decreasing returns to scale The production function has constant returns to scale The production function has increasing returns to scale Marginal returns are decreasing Marginal returns are constant Marginal returns are increasing It implies that factor shares are decreasing It implies that factor shares are constant It implies that factor shares are increasing 2 1 point. Which of the following production functions satisfy all the desired properties from the previous question? F(K, L) AK0.3 0.3 F(K, L) = AKL F(K,L) AKL¹-a F(K,L) AK0-3L0.7 F(K, L) = AK + L 000000000000 Which of the following are desired properties of the aggregate production function? (Make sure you understand the reason why these are desired properties) Output is decreasing in all its factors Output is constant in all its factors Output is increasing in all its factors. The production function has decreasing returns to scale The production function has constant returns to scale The production function has increasing returns to scale Marginal returns are decreasing Marginal returns are constant Marginal returns are increasing It implies that factor shares are decreasing It implies that factor shares are constant It implies that factor shares are increasing 2 1 point. Which of the following production functions satisfy all the desired properties from the previous question? F(K, L) AK0.3 0.3 F(K, L) = AKL F(K,L) AKL¹-a F(K,L) AK0-3L0.7 F(K, L) = AK + L 000000000000
Expert Answer:
Answer rating: 100% (QA)
Explanation 1 Output is increasing in all its factors Upon i... View the full answer

Related Book For 

Principles of Auditing and Other Assurance Services
ISBN: 978-0078025617
19th edition
Authors: Ray Whittington, Kurt Pany
Posted Date:
Students also viewed these chemical engineering questions
-
Which of the following are Section 1231 assets? Explain. Assume all the items have been held long-term. a. Machinery used in the business b. Personal home c. Factory building d. Land held as an...
-
Which of the following are private goods and might, therefore, be provided in socially optimal amounts by private profit-maximizers? Which are public goods and should, therefore, be provided by the...
-
Which of the following are probability distributions? Why? (a) RANDOM VARIABLE X PROBABILITY 2 ................... 0.1 1 ................. 0.2 0 ................... 0.3 1 ................... 0.25 2...
-
Engineers observe that about 90% of graphite samples fracture within five hours when subjected to a certain stress. (a) If the time to fracture is modeled with an exponential distribution, what would...
-
Harlen Industries has a simple forecasting model: Take the actual demand for the same month last year and divide that by the number of fractional weeks in that month. This gives the average weekly...
-
In the book Cases in Finance, Nunnally and Plath present a case in which the estimated percentage of uncollectible accounts varies with the age of the account. Here the age of an unpaid account is...
-
When a party may be compelled to defend an action in a particular court?
-
A privately owned summer camp for youngsters has the following data for a 12-week session: Charge per camper ................................$120 per week Fixed costs...
-
A seller of real estate, who does not have a real estate license, swindles a buyer out of $60,000. How much, at a maximum, might the buyer recover from Indiana Real Estate Recovery Fund?
-
SECTION B: ANSWER ANY THREE (3) QUESTIONS QUESTION TWO The following summarized statements of comprehensive income have been prepared for the year ended 31 December 2014 for Gold Reef Holdings and...
-
A RISC processor is designed to have following instruction types. It is decided to process instructions in five sub-tasks/steps, in general and processing circuits of different sub-tasks require...
-
Blue Computer had the following transactions during the month of May. 1. Purchased equipment on account for $9,700. 2. Paid $1,500 for rent for the month of May. 3. Performed computer services for...
-
what is the risk management? Explain
-
Identify the correct format for an APA reference-list entry for an article in a scholarly journal. The article is by Yao Lu and is titled "Empowerment or Disintegration? Migration, Social...
-
explain the role of Value at Risk (VaR) in managing the risk of complex financial portfolios, and what are its limitations?
-
The kinetics of the polymerization of methyl methacrylate monomer were studied at 77 C using benzene as a solvent and azo-bisisobutylronitrile (AIBN) as the free radical initiator. The following...
-
In his seminal article The Necessary and Sufficient Conditions of Therapeutic Personality Change, originally published in 1957, Rogers identified three core components of what he considered to be the...
-
Chloroplasts are illuminated until the levels of the Calvin cycle intermediates reach a steady state. The light is then turned off. How does the level of RuBP vary after this point?
-
The clients cost accounting system is often an important part of the CPAs audit of the financial statements of a manufacturing company. For what purposes do the auditors consider the cost accounting...
-
List three substantive procedures the auditors could use to detect unrecorded retirements of property, plant, and equipment.
-
Assume that CPAs are attesting to comparative financial statements. Can the CPAs express differing opinions on the sets of financial statements of two successive years?
-
When there is a concentration gradient in the system, show that the potential gradient is composed of two terms, (i) an Ohm's-law contribution and (ii) a diffusional contribution. State the equation...
-
Copper is deposited at a cathode from solution with a bulk concentration of \(0.5 \mathrm{M}\) at the rate of \(3.0 \mathrm{~g} / \mathrm{m}^{2} \cdot \mathrm{s}\). Find the surface concentration of...
-
Find the mobility of \(\mathrm{H}^{+}, \mathrm{OH}^{-}\), and other ions from the diffusivity data given in Table 22.1. Table 22.1. Diffusion coefficients of ions in water at 25 C. H+ Na+ K+ Ca+ OH-...

Study smarter with the SolutionInn App


