Which of the following contain/s polar 1 point bonds but overall, is/are a nonpolar molecule? *...
Fantastic news! We've Found the answer you've been seeking!
Question:


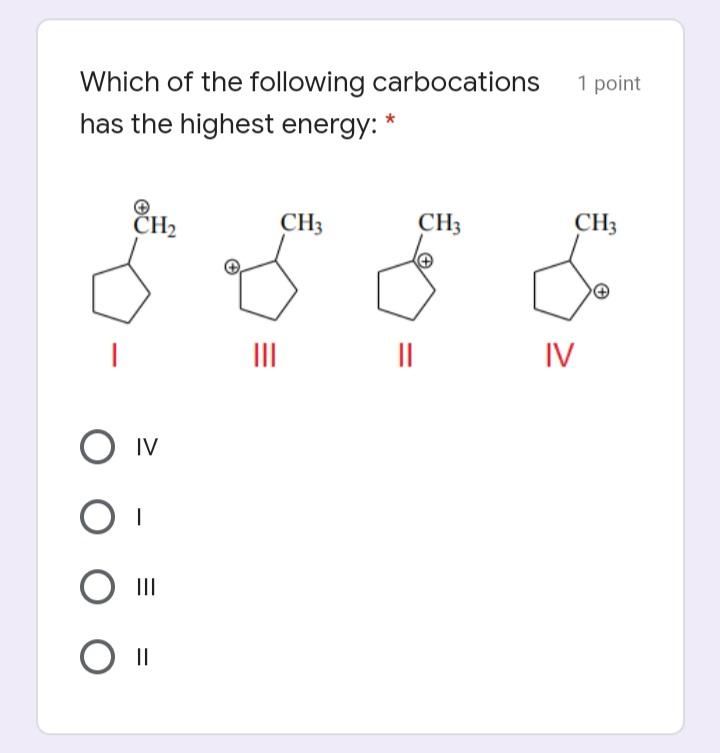
Transcribed Image Text:
Which of the following contain/s polar 1 point bonds but overall, is/are a nonpolar molecule? * PCI5 CO2 PC13 NC13 Arrange the following in decreasing 1 point order with respect to relative acidity of the highlighted H atom in each of the following: ~pKa: terminal alkyne = 25, ammonia = 35, water = 15) * НО-Н CH3CEC-H H2N-H i ii i i > iii > ii i> ii > iii iii > i > i O ii > i > ii Which of the following is/are both a 1 point Lewis Acid and Brønsted Acid? Please select * А) Н-РО В) РО,3- С) NНа D) F- E) BF; C E A D Which of the following carbocations has the highest energy: * 1 point CH2 CH3 CH3 CH3 IV O IV II Which of the following contain/s polar 1 point bonds but overall, is/are a nonpolar molecule? * PCI5 CO2 PC13 NC13 Arrange the following in decreasing 1 point order with respect to relative acidity of the highlighted H atom in each of the following: ~pKa: terminal alkyne = 25, ammonia = 35, water = 15) * НО-Н CH3CEC-H H2N-H i ii i i > iii > ii i> ii > iii iii > i > i O ii > i > ii Which of the following is/are both a 1 point Lewis Acid and Brønsted Acid? Please select * А) Н-РО В) РО,3- С) NНа D) F- E) BF; C E A D Which of the following carbocations has the highest energy: * 1 point CH2 CH3 CH3 CH3 IV O IV II
Expert Answer:
Answer rating: 100% (QA)
1 CO2 it has polar CO bonds but due to symmetrical ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A rate law is one-half order with respect to a reactant. What is the effect on the rate when the concentration of this reactant is doubled?
-
Select the best answer. 1. Which of the following would be least likely to be classied as a citys general capital assets? a. Roads and bridges b. Electric utility lines c. Computers used by the...
-
Which of the following are positive incentives? Negative incentives? Why? a. A fine for not cleaning up after your dog defecates in the park b. A trip to Hawaii paid for by your parents or...
-
Evaluate the limit or determine that it does not exist. |xx| lim (x,y)(0,0) [x] + [yl
-
What factors may affect the availability of the remedy of rescission?
-
A slipper-pad bearing (Fig. P1023) is often encountered in lubrication problems. Oil flows between two blocks; the upper one is stationary, and the lower one is moving in this case. The drawing is...
-
Why has the importance of environmental accounting to business grown in the past ten years?
-
1. Do you agree with Cairnss assertion that classifications of accounting are simplistic and of little relevance in todays world? Are attempts to classify accounting futile and outmoded? Why or why...
-
On successful completion of MBA, you have joined a human resource consultant firm. You have been asked to prepare the job descriptions for the following category of organizations (Job titles...
-
A column fixed at both ends has a minimum radius of gyration r = 29.4 mm, cross-sectional area 3,230 mm, and length 1.94 m. It is made of a material whose compression stress-strain diagram may be...
-
In an experiment, m grams of a compound X (gas/liquid/solid) taken in a container is loaded in a balance as shown in figure I below. In the presence of a magnetic filed, the pan with X is either...
-
How does Praxis integrate Agile into its project management approach?
-
What is the significance of identifying long lead-time items and large capital items early on in the project life cycle?
-
What additional information can be gained from simple spreadsheet budgets to monitor and control project costs? Remember the 'planned' and 'actual', and tallies across the rows.
-
What are the advantages of Agile? What are the disadvantages of Agile?
-
What type of project(s) is the Lean Six Sigma approach best suited for?
-
From where can the variable x be accessed in this Java code? public class Test { int x; public Test() { } // A public int doStuff() { int y; // B for (int i = 0; i < 10; i++) { int = 3; // C } public...
-
Place a tick in the appropriate grid to identify the balance that would be brought down in each of the following named accounts, in the books of Rizwy Mohamed: (a) In the Cash account: if Rizwy...
-
How many moles of calcium chloride, CaCl2, can be added to 1.5 L of 0.020 M potassium sulfate, K2SO4, before a precipitate is expected? Assume that the volume of the solution is not changed...
-
Consider a reaction in which Ho and So are positive. Suppose the reaction is nonspontaneous at room temperature. How would you estimate the temperature at which the reaction becomes spontaneous?
-
Explain why barium fluoride dissolves in dilute hydrochloric acid but is insoluble in water.
-
a. What responsibility does the auditor have when he believes material errors or irregularities may exist? b. What are the possible effects of the foregoing on the auditor's standard report?
-
Watts and Williams, a firm of certified public acccountants, audited the accounts of Sampson Skins, Inc., a corporation that imports and deals in fine furs. Upon completion of the examination the...
-
a. Can an examination made in accordance with generally accepted auditing standards be relied upon to detect illegal acts? Why or why not? b. What are the possible effects of illegal acts on the...

Study smarter with the SolutionInn App


