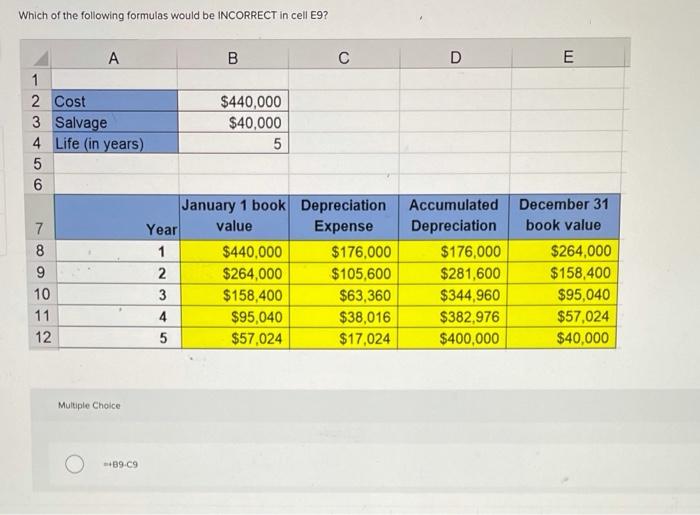
Which of the following formulas would be INCORRECT in cell E9? C D E 1 2...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
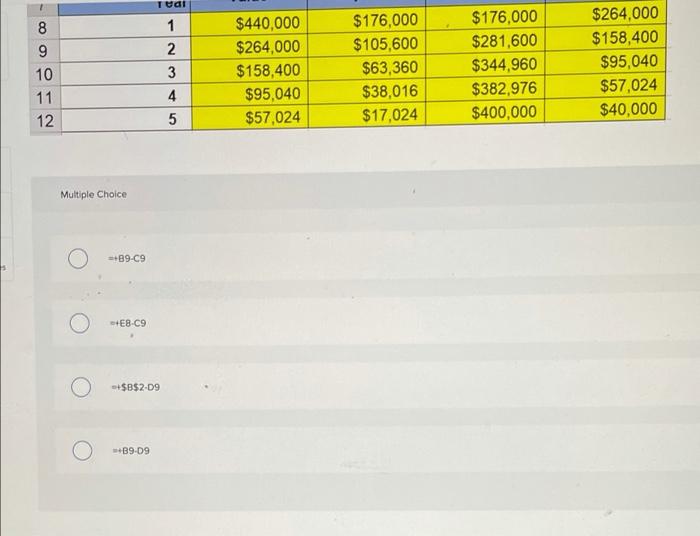
Which of the following formulas would be INCORRECT in cell E9? C D E 1 2 Cost 3 Salvage 4 Life (in years) $440,000 $40,000 January 1 book Depreciation Year Accumulated December 31 7 value Expense Depreciation book value 8 1 $440,000 $176,000 $176,000 $264,000 $264,000 $105,600 $281,600 $158,400 10 3 $158,400 $63,360 $344,960 $95,040 11 4 $95,040 $38,016 $382,976 $57,024 12 $57,024 $17,024 $400,000 $40,000 Multiple Choice +89-C9 56 $440,000 $176,000 $176,000 $264,000 1 $264,000 $105,600 $281,600 $158,400 2 $158,400 $63,360 $344,960 $95,040 10 3 $95,040 $38,016 $382,976 $57,024 11 4 $57,024 $17,024 $400,000 $40,000 12 Multiple Choice =+89-C9 +E8-C9 +$B$2-D9 +B9-D9 5 8. Which of the following formulas would be INCORRECT in cell E9? C D E 1 2 Cost 3 Salvage 4 Life (in years) $440,000 $40,000 January 1 book Depreciation Year Accumulated December 31 7 value Expense Depreciation book value 8 1 $440,000 $176,000 $176,000 $264,000 $264,000 $105,600 $281,600 $158,400 10 3 $158,400 $63,360 $344,960 $95,040 11 4 $95,040 $38,016 $382,976 $57,024 12 $57,024 $17,024 $400,000 $40,000 Multiple Choice +89-C9 56 $440,000 $176,000 $176,000 $264,000 1 $264,000 $105,600 $281,600 $158,400 2 $158,400 $63,360 $344,960 $95,040 10 3 $95,040 $38,016 $382,976 $57,024 11 4 $57,024 $17,024 $400,000 $40,000 12 Multiple Choice =+89-C9 +E8-C9 +$B$2-D9 +B9-D9 5 8.
Expert Answer:
Answer rating: 100% (QA)
Solution Book value can be computed as in follwing ways 1 Book value on Jan... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Which of the following formulas for (f, g) define inner products on the space C°[ - 1, 1 ]? a. b. c. d. Fee f(x)g(x)e dx f(x) g(x)x dx f (x)g(x) (x + 2) dx f(x)g(x)x2 dx
-
Multiple - Choice Questions 1. Which of the following is not a specific account in a companys chart of accounts? a. Gains b. Revenue c. Net Income d. Unearned Revenue 2. Which of the following is not...
-
Multiple Choices Questions 1. Which of the following would be considered a significant deficiency in an organization's control environment? a. The internal audit function is outsourced to a public...
-
A group of 70 students were asked to record the last digit of their social security number. a. Draw a relative frequency histogram using the values 0 through 9 as the class midpoints. What is the...
-
Refer to the information in E109. Power Drive Corporation has the following beginning balances in its stockholders equity accounts on January 1, 2015: Common Stock, $200,000; Additional Paid-in...
-
If XYZ does nothing to manage copper price risk, what is its profit 1 year from now, per pound of copper? If on the other hand XYZ sells forward its expected copper production, what is its estimated...
-
Although physically counting a businesss inventory is integral to the periodic system of inventory recordkeeping, it is not an explicit component of the perpetual system. Nonetheless, most well-run...
-
Janet Hardee runs a popcorn stand in the local amusement park. Her cost to produce a bag of popcorn is $0.40, which she sells for $2.00. Required a. What is Janet's markup percentage on a bag of...
-
1. Identily how the following situation would affect an auditors risk analysis for a company should include the following: Identify how the situation affects overall Audit Risk (AR) - that is, is the...
-
Question 3b (10 points): Apply Linear Regression to predict call volume from head count using the appropriate Excel template (Linerar_Regression Model.xlsx). Based on the upcoming acquisition of...
-
What is the importance of theoretical flame temperature?
-
Carbon dioxide occupies a tank at \(100^{\circ} \mathrm{C}\). If the volume of the tank is \(0.5 \mathrm{~m}^{3}\) and the pressure is \(500 \mathrm{kPa}\), determine the mass of the gas in the tank.
-
What is the difference between excess air and theoretical air?
-
Give an informatory note on saturation temperature and saturation pressure.
-
Name the factors responsible for incomplete combustion even if excess air is supplied to the fuel.
-
TTL is the abbreviation for through the lens. Question 24 Answer a. True b. False
-
What mass of H2 will be produced when 122 g of Zn are reacted? Zn(s) + 2HCl(aq) ( ZnCl2(aq) + H2(g)
-
Suppose the bottom support in the mass-spring chain in Example 9.36 is removed. (a) Do you predict that the vibration rate will (i) Speed up. (ii) Slow down, or (iii) Stay the same? (b) Verify your...
-
Verify the triangle inequality for the vectors and inner products in Exercise 3.2.4.
-
Find the eigenvalues, to 2 decimal places, of the matrices in Exercise 10.6.18 by applying the QR algorithm to the tridiagonal form. In Exercise 10.6.18 Use Householder matrices to convert the...
-
Discuss what is meant by the tone at the top and why it is important. Further, discuss whether you feel it is ethical to be a whistle-blower.
-
Consider the following independent situations: 1. An international health club sells lifetime memberships costing \($1,500\) which allow the purchaser unlimited use of any of the clubs 300 facilities...
-
The Longo Corporation contracted with The Davis Company to manufacture various metal component parts that would be assembled by Longo before resale to Longos customers. Longo placed its most recent...

Study smarter with the SolutionInn App


