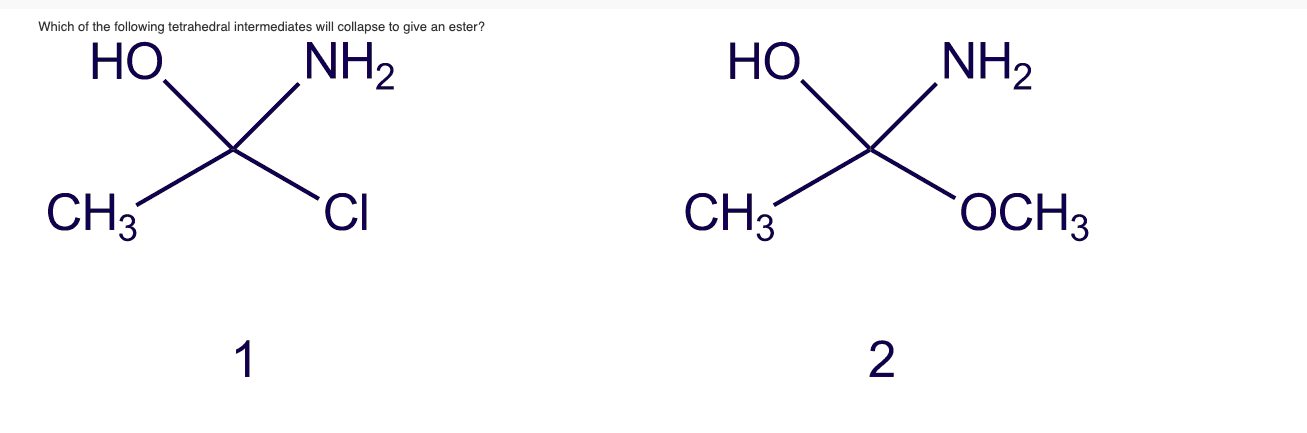
Which of the following tetrahedral intermediates will collapse to give an ester? HO NH CH3 1...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
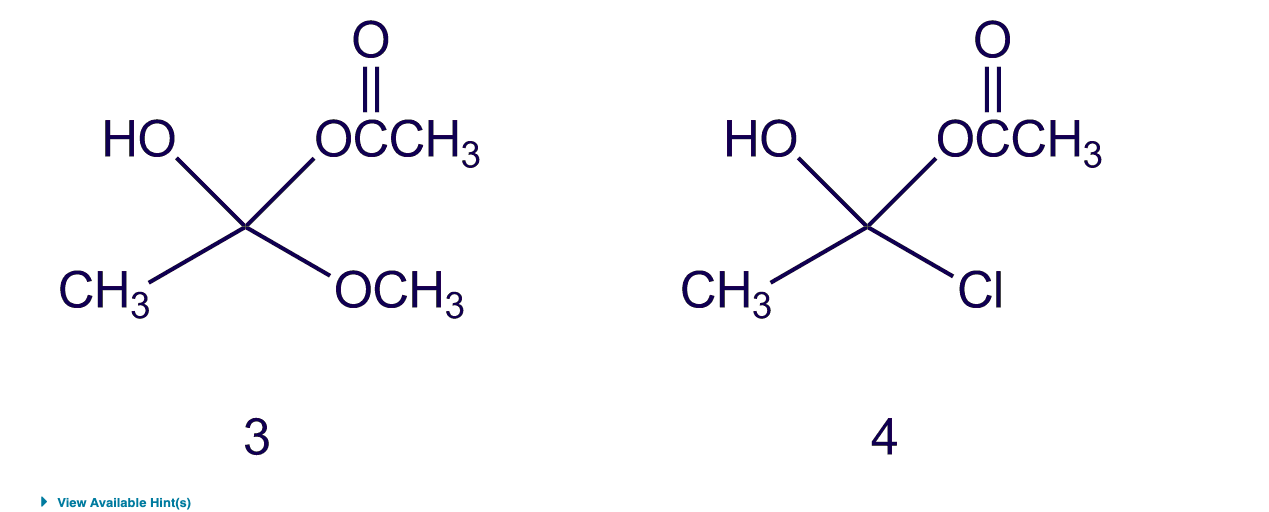
Which of the following tetrahedral intermediates will collapse to give an ester? HO NH₂ CH3 1 CI HO CH3 2 NH₂ OCH 3 HO CH3 ► View Available Hint(s) 3 O OCCH3 OCH 3 HO CH3 4 O OCCH3 CI View Available Hint(s) O 3 02 O 1 Submit Which of the following tetrahedral intermediates will collapse to give an ester? HO NH₂ CH3 1 CI HO CH3 2 NH₂ OCH 3 HO CH3 ► View Available Hint(s) 3 O OCCH3 OCH 3 HO CH3 4 O OCCH3 CI View Available Hint(s) O 3 02 O 1 Submit
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Which of the following compounds would give a positive Tollens test? (Remember that the Tollens test involves mild basic aqueous conditions.) (a) CH3CH2CH2COCH3 (b) CH3CH2CH2CH2CHO (c)...
-
Which of the following would be expected to give a positive test with Benedict's reagent? Why? (a) L-Arabinose (b) 1,3-Dihydroxyacetone (c) D-Fructose (d) Lactose (e) Amylose
-
Which of the following 2 x 2 matrices are positive definite? (a) (b) (c) (d) (e) (f) In the positive definite cases, write down the formula for the associated inner product. 0 2 2 1 2 2 3 (1 -3)
-
For the following exercises, use shells to find the volume generated by rotating the regions between the given curve and y = 0 around the x-axis. 130. y = 1-x,x = 0, and x = 1 131. y = x, x = 0, and...
-
Calculate the normal time for each of the workelements. Mean Performance Rating Factor 0.95 0.85 1.10 1.10 Work Observed Time Element (minutes) 1.20 1.00 0.80 0.90
-
Chris North is the founder and president of North Enterprises, a real estate development venture. The business transactions during April while the company was being organized are listed below. Apr. 1...
-
Assuming the same data as given in problem 9, was the well in each case profitable? Discuss your answer. Problem 9:- Property cost (acquisition cost). Drilling cost (one well). Estimated completion...
-
The Marlin Company produces plastic bottles to customer order. The quality inspector randomly selects four bottles from the bottle machine and measures the outside diameter of the bottle neck, a...
-
An aluminum flagpole is 34 m high. By how much does its length increase as the temperature increases by 23 C? (The linear expansion coefficient of aluminum is 23 x 10-6 /C)
-
Starting with the finished version of Example 9.3, change the fixed marketing cost to $4000 (really $4 million) in cell B5, and change the decision criterion to maximize expected utility, using an...
-
(a) From the following information, prepare a Stores Ledger Account under First-In-First Out (FIFO) method : 2015 Jan.1 Jan. 10 Jan. 12 Jan. 22 Jan. 25 Jan. 31 Ans. Opening balance 1000 units @ *...
-
Provided $17,300 in animal care services for customers on April 3, all on credit. What accounts would this effect?
-
Discuss the concept of double-entry accounting and its benefits. What is the significance of the materiality principle in accounting standards?
-
Discuss any five (5) tools that can be used to recover deleted files and five (5) tools used in digital forensic and Discuss the advantages and the contributions of cyber security in our everyday...
-
Using a factory overhead allocation method such as the single plantwide factory overhead rate can cause product cost distortions. However, this method is less complicated and inexpensive to maintain....
-
Both financial and managerial accounting are important to a company's success. What do you think contributes more to this success and why?
-
If you were the front office manager, what would you do to solve the immediate problems? After the commotion had settled down, how would you analyze the situation? List the opportunities for...
-
In the synthesis of the keto acid just given, the dicarboxylic acid decarboxylates in a specific way; it gives Explain. HO rather than HO
-
Rank the following compounds in order of decreasing ease of removing a proton from a methyl group: CH3 CH3 CH3 CH2CH3
-
What carbonyl compound and what phosphonium ylide are needed to synthesize the following compounds? a. b. c. d. CH-CHCH2CH2CH3 CHCH2CH, CH CH CH2
-
Account for the difference in the shape and color of the potential maps for ammonia and the ammonium ion in Section 1.12.
-
The SDLC is just one model for systems development. Find at least one more and describe the differences.
-
Draw DFDs for each of these scenarios: (a) A customer goes into a bookshop and asks for this book. The member of staff looks for the book in the online stock catalogue and reports that the book is...
-
Draw an entity model to model this university scenario: A university department employs lecturers and clerical staff. It offers a three-year degree. A student has to take 12 modules during the...

Study smarter with the SolutionInn App



