Question: which one is the corresponding Maxwell relationship derived from dH=TdS+VdP a. b, dP ()s - ($)P ()s--(%)r Od. 6P
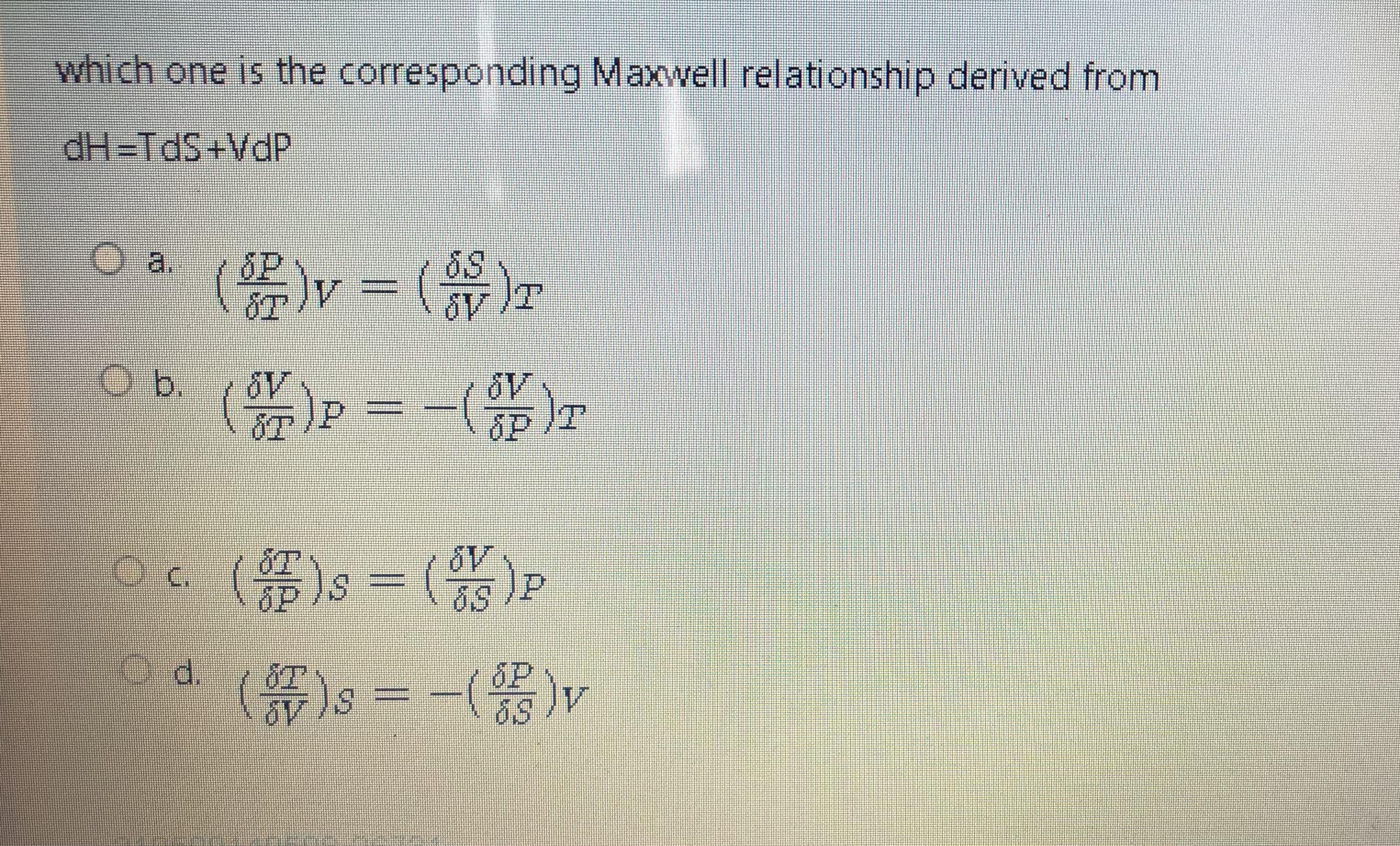
which one is the corresponding Maxwell relationship derived from dH=TdS+VdP a. b, dP ()s - ($)P ()s--(%)r Od. 6P
Step by Step Solution
3.37 Rating (153 Votes )
There are 3 Steps involved in it
C ... View full answer

Get step-by-step solutions from verified subject matter experts


