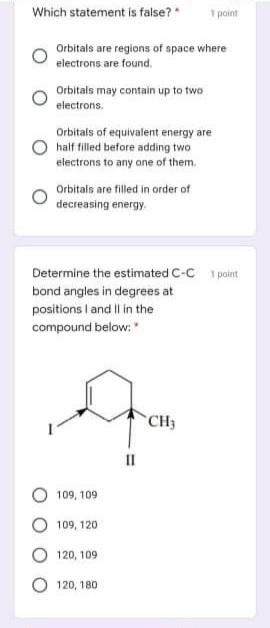
Which statement is false? 1 point Orbitals are regions of space where electrons are found, Orbitals...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
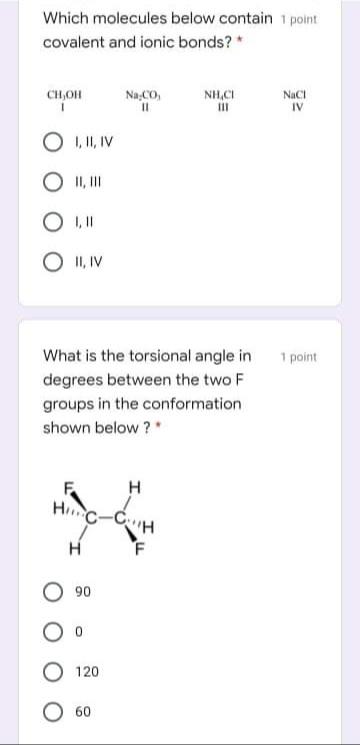
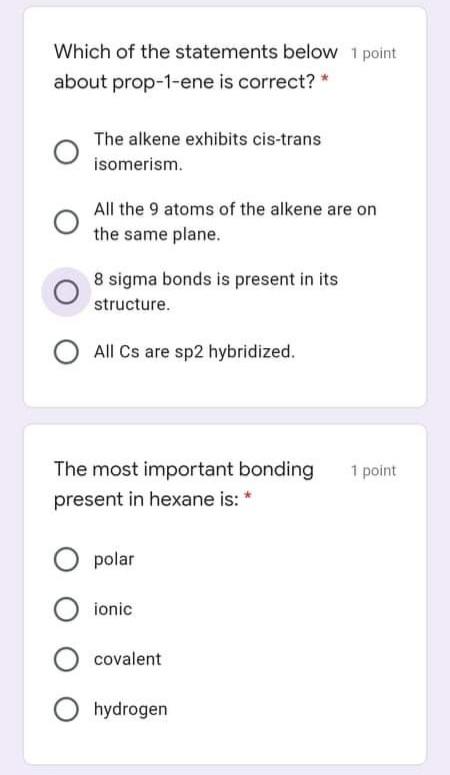
Which statement is false? 1 point Orbitals are regions of space where electrons are found, Orbitals may contain up to two electrons. Orbitals of equivalent energy are half filled before adding two electrons to any one of them. Orbitals are filled in order of decreasing energy. Determine the estimated C-C 1 point bond angles in degrees at positions I and Il in the compound below: CH3 II 109, 109 109, 120 120, 109 O 120, 180 Which molecules below contain 1 point covalent and ionic bonds? * Na CO, %3D NH,CI CH,OH NaCI IV O I, 1, IV O I,II O II, IV 1 point What is the torsional angle in degrees between the two F groups in the conformation shown below ?" H C-C "H F 90 О 120 O 60 Which of the statements below 1 point about prop-1-ene is correct? * The alkene exhibits cis-trans isomerism. All the 9 atoms of the alkene are on the same plane. 8 sigma bonds is present in its structure. All Cs are sp2 hybridized. The most important bonding 1 point present in hexane is: * O polar O ionic covalent O hydrogen Which of the following 1 point statements are correct? Conformation exist in a dynamic equilibrium state. Conformers cannot be separated. Conformers differ largely in energies. Amount of more stable conformers is more than that of less stable conformers at equilibrium. Which of the following is not 1 point true? * Delocalized electrons destabilize a compound. O All options are not true. Resonance is the extra stability a compound possesses from having delocalized electrons. The greater the number of stable resonance contributors, the greater the resonance stabilization is Which statement is false? 1 point Orbitals are regions of space where electrons are found, Orbitals may contain up to two electrons. Orbitals of equivalent energy are half filled before adding two electrons to any one of them. Orbitals are filled in order of decreasing energy. Determine the estimated C-C 1 point bond angles in degrees at positions I and Il in the compound below: CH3 II 109, 109 109, 120 120, 109 O 120, 180 Which molecules below contain 1 point covalent and ionic bonds? * Na CO, %3D NH,CI CH,OH NaCI IV O I, 1, IV O I,II O II, IV 1 point What is the torsional angle in degrees between the two F groups in the conformation shown below ?" H C-C "H F 90 О 120 O 60 Which of the statements below 1 point about prop-1-ene is correct? * The alkene exhibits cis-trans isomerism. All the 9 atoms of the alkene are on the same plane. 8 sigma bonds is present in its structure. All Cs are sp2 hybridized. The most important bonding 1 point present in hexane is: * O polar O ionic covalent O hydrogen Which of the following 1 point statements are correct? Conformation exist in a dynamic equilibrium state. Conformers cannot be separated. Conformers differ largely in energies. Amount of more stable conformers is more than that of less stable conformers at equilibrium. Which of the following is not 1 point true? * Delocalized electrons destabilize a compound. O All options are not true. Resonance is the extra stability a compound possesses from having delocalized electrons. The greater the number of stable resonance contributors, the greater the resonance stabilization is
Expert Answer:
Answer rating: 100% (QA)
1 the orbital is defined as it is the threedimensional space around the nucleus where the probabilit... View the full answer

Related Book For 

Applied Statistics in Business and Economics
ISBN: 978-0073521480
4th edition
Authors: David Doane, Lori Seward
Posted Date:
Students also viewed these chemistry questions
-
Which statement is false concerning reporting standards for these investments Select one A. Interest received on trading and AFS debt investments reduces the investment balance B. Fizzy reports...
-
For a multiple regression, which statement is false ? Explain. a. If R2 = .752 and R2adj = .578, the model probably has at least one weak predictor. b. R2adj can exceed R2 if the model contains some...
-
Which compound has bond angles nearest to 120? (a) O = C = S (b) CHI3 (c) H2C = O (d) H-C=C-H (e) CH4
-
1. Given the image of the circle, identify the length of the radius, diameter, and circumference. Also, find the area. Make sure to show work (area and circumference) to receive credit. Choose a...
-
Make an amortization table to show the first two payments for the mortgages in Exercise 1 - 6. See Example 1. Monthly interest rate Years payment $1,022.40 Monthly Amount of mortgage 1. $100,000...
-
Discuss one advantage and one disadvantage to the client of each managers contracted fee structure. Institutional investment consultant Wilsot Consultants (Wilsot) is reviewing multiple investment...
-
For the quarter domain of an RVE in Figure 3.23, a uniform transverse normal stress \(\bar{\sigma}_{x}\) is applied on the plane \(x=D / 2\). Set up the equations describing the boundary conditions...
-
1. What are some of the current issues facing Germany? What is the climate for doing business in Germany today? 2. Are the leadership styles used by the German managers really much different from...
-
What increases brain activity and how does it relate to what is consciousness? What leads to the loss of consciousness? illustrate the process of consciousness? is consciousness gradual? Explain. How...
-
Trace or copy the graph of the given function f. (Assume that the axes have equal scales.) Then use the method of Example 1 to sketch the graph of f' below it. (a) (b) (c) yA
-
Complete all parts listed under the Concepts section and the Process section as given in the case. For the ANALYSIS Section: Complete all three tables in part (f) as given. New: (f) i. of the values...
-
Use the tables on the page for the following problems. What would a 150/300/75 liability insurance semiannual premium be in territory 4? If you were a 21 year old male who lives in territory 2...
-
Answer the following numerical questions based on the 1. What is the mean value of the resistance of the set of resistors? 2. What is the standard deviation of the resistance of the set of resistors?...
-
George and Martha Washington ask you to prepare their tax return for 2018. They give you the following information upon which to work. 1) George's social security number is 123-45-6789, while...
-
Superior Limited's total overhead costs at various levels of activity are presented below: Month March April May June Machine Hours Total Overhead Costs 60,000 $216,800 50,000 194,000 70,000 239,600...
-
The tab contains monthly holding period stock returns for International Business Machines Corp. (IBM), Microsoft Corp. (MSFT), and Caterpillar Inc. (CAT) for the months from January 2016 through...
-
Anthem's avoidable emergency room policyAnthem, one of the largest healthcare insurers in the United States, implemented an "avoid-able ER" policy to help manage the care of its enrollees. The policy...
-
Illini Company, Inc. Balance Sheet as of 12/31/20X0 Assets Current Assets: Cash $1,500,000 Accounts receivable, net 18,000 Inventory 50,000 Total current assets 1,568,000 Equipment 90,000 Goodwill...
-
Several friends go bowling several times per month. They keep track of their scores over several months. An ANOVA was performed. (a) What kind of ANOVA is this (one-factor, two-factor, etc.)? (b) How...
-
Given P (A) = .40, P (B) = .50. If A and B are independent, find P (A B).
-
For the following contingency table, find (a) P(H © T): (b) P (S | G): (c) P (S) 10 20 30 50 50 100 30 40 70 Row Total 90 110 200 Col Total
-
T/F: BOs are internally stable and externally adaptable.
-
T/F: BOs is the middle layer of stability model.
-
T/F: BOs are semi-tangible and mostly conceptual.

Study smarter with the SolutionInn App


