Whitman Company has just completed its first year of operations. The company's absorption costing income statement...
Fantastic news! We've Found the answer you've been seeking!
Question:
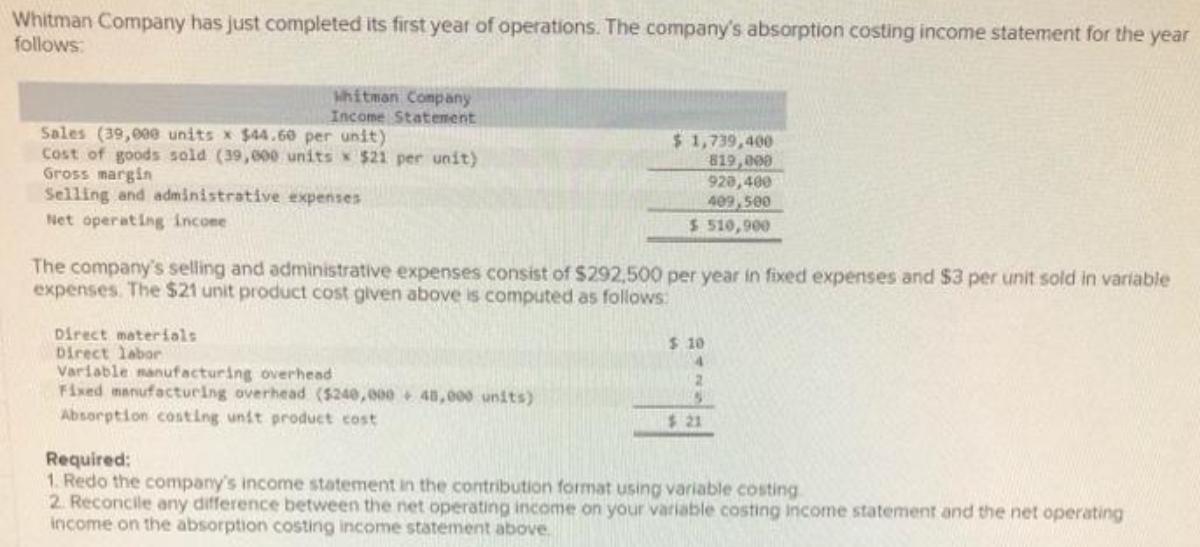
Transcribed Image Text:
Whitman Company has just completed its first year of operations. The company's absorption costing income statement for the year follows: Whitman Company Income Statement Sales (39,000 units x $44.60 per unit) $1,739,400 Cost of goods sold (39,000 units $21 per unit) Gross margin 819,000 920,400 409,500 Selling and administrative expenses Net operating income $ 510,900 The company's selling and administrative expenses consist of $292,500 per year in fixed expenses and $3 per unit sold in variable expenses. The $21 unit product cost given above is computed as follows: Direct materials Direct labor $ 10 4 Variable manufacturing overhead 2 Fixed manufacturing overhead ($240,000+ 48,000 units) Absorption costing unit product cost $21 Required: 1. Redo the company's income statement in the contribution format using variable costing 2. Reconcile any difference between the net operating income on your variable costing Income statement and the net operating income on the absorption costing income statement above Whitman Company has just completed its first year of operations. The company's absorption costing income statement for the year follows: Whitman Company Income Statement Sales (39,000 units x $44.60 per unit) $1,739,400 Cost of goods sold (39,000 units $21 per unit) Gross margin 819,000 920,400 409,500 Selling and administrative expenses Net operating income $ 510,900 The company's selling and administrative expenses consist of $292,500 per year in fixed expenses and $3 per unit sold in variable expenses. The $21 unit product cost given above is computed as follows: Direct materials Direct labor $ 10 4 Variable manufacturing overhead 2 Fixed manufacturing overhead ($240,000+ 48,000 units) Absorption costing unit product cost $21 Required: 1. Redo the company's income statement in the contribution format using variable costing 2. Reconcile any difference between the net operating income on your variable costing Income statement and the net operating income on the absorption costing income statement above
Expert Answer:
Related Book For 

Managerial Accounting
ISBN: 978-0697789938
13th Edition
Authors: Ray H. Garrison, Eric W. Noreen, Peter C. Brewer
Posted Date:
Students also viewed these accounting questions
-
Whitman Company has just completed its first year of operations. The companys absorption costing income statement for the year appears below: The companys selling and administrative expenses consist...
-
The Bradley Company has just completed its first year of operations. A condensed income statement follows, showing actual and standard amounts and the variances: Required: The president of Bradley...
-
Classify each of the following items as a public good, a private good, a mixed good, or a common resource. Georges Banks cod stock A courtside seat at the U.S. Open (tennis) A well-stocked buffet...
-
The GASB requires a government to prepare budgetary comparisons for its general fund and major special revenue funds that have a legally adopted annual budget. The city for which you work does not...
-
Given the following two classes in the same package, what is the result of executing the Hug program? A. kitty - 5.0 B. The Kitten class does not compile. C. The Hug class does not compile. D. The...
-
Calculate the plate temperature in Example 7.3 at 10 a.m., if the insolation during the first \(3 \mathrm{~h}\) is 0,150 , and \(270 \mathrm{~W} / \mathrm{m}^{2}\), respectively, and the air...
-
Renkas Heaters selected data for October 2011 are presented here (in millions): Direct materials inventory 10/1/2011 $ 105 Direct materials purchased 365 Direct materials used 385 Total manufacturing...
-
A battery of 12 V is connected across capacitors of capacitances C1-C2=C3 C4 =C7= B F, Cs = C6= F. 12 V C3 C2 C1 i. What is the equivalent capacitance Ceq of the capacitors, you should show your work...
-
Recording a Business Combination Petra Corporation paid $ 8 million in cash to acquire the assets and liabilities of Salisbury Company. Petra also agreed to make an additional cash payment in the...
-
For a number of years, a private not-for-profit entity has been preparing financial statements that do not necessarily follow generally accepted accounting principles. At the end of the most recent...
-
Describe the core elements required within the DSCSA (Drug Supply Chain Security Act) Describe the role of pharmacy benefit managers (PBMs) in the US Healthcare System Explain the overall structure...
-
Discuss the significance of Process Hazard Analysis (PHA) techniques, such as Hazard and Operability Studies (HAZOP) and Failure Modes and Effects Analysis (FMEA), in identifying potential risks in...
-
Joe runs up the stadium steps as an athletic drill. There are 50 steps and each one is 0.30 meters high and 0.50 meters deep. If Joe has a mass of 95 kg and runs the steps in 30 seconds, how much...
-
You are an investigator called to the scene of a suspicious death. Once on scene, you are told by the initial responding patrol officer that a relative of the female occupant of the home came to the...
-
How does the concept of inherently safer design (ISD) integrate with hazard analysis to promote safety at the earliest stages of process design? Provide examples where ISD principles have...
-
What Do Entrepreneurs Do? assesses the potential for the entrepreneurial venture launches the venture organizes the venture All of the above
-
What is the maximum volume of 0.25 M sodium hypochlorite solution (NaOCl, laundry bleach) that can be prepared by dilution of 1.00 L of 0.80 M NaOCl?
-
Han Products manufactures 30,000 units of part S-6 each year for use on its production line. At this level of activity, the cost per unit for part S-6 is as follows: An outside supplier has offered...
-
Feather Friends, Inc., distributes a high-quality wooden birdhouse that sells for $20 per unit. Variable costs are $8 per unit, and fixed costs total $180,000 per year. Required: Answer the following...
-
Lubricants, Inc., produces a special kind of grease that is widely used by race car drivers. The grease is produced in two processing departments: Refining and Blending. Raw materials are introduced...
-
Show that \[ \gamma_{i}=\frac{\bar{\phi}_{i}}{\phi_{i}} \]
-
Carbon dioxide occupies a tank at \(100^{\circ} \mathrm{C}\). If the volume of the tank is \(0.5 \mathrm{~m}^{3}\) and the pressure is \(500 \mathrm{kPa}\), determine the mass of the gas in the tank.
-
Give an informatory note on saturation temperature and saturation pressure.

Study smarter with the SolutionInn App



