6. A 125 mL portion of pure water is mixed with 175 mL of 0.120 M...
Fantastic news! We've Found the answer you've been seeking!
Question:
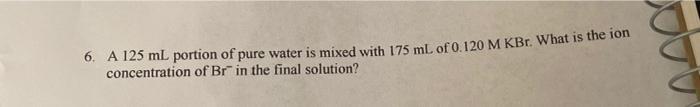
Transcribed Image Text:
6. A 125 mL portion of pure water is mixed with 175 mL of 0.120 M KBr. What is the fon concentration of Br in the final solution? 6. A 125 mL portion of pure water is mixed with 175 mL of 0.120 M KBr. What is the fon concentration of Br in the final solution?
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
At 27C, the vapor pressure of pure water is 23.76 mmHg and that of an urea solution is 22.98 mmHg. Calculate the molality of solution.
-
At 25C the vapor pressure of pure water is 23.76 mmHg and that of seawater is 22.98 mmHg. Assuming that seawater contains only NaCl, estimate its molal concentration.
-
1. We learned that the pH of pure water is 7. How many H30+ molecules are present in 1 liter of water? 3 pts Hint: You can assume that all H + have associated with H 2 0
-
Transactions related to revenue and cash receipts completed by Acheville Architects Co. during the period September 2-30, 2014, are as follows: Sept. 2. Issued Invoice No. 793 to Nickle Co., $5,200....
-
Indicate whether each of the following statements is true or false by writing T or F in the answer column. 1. While the term malpractice is usually applied to physicians, dentists, attorneys, and...
-
(a) Use the analogy between Faraday's law and Ampere's law, together with the Biot-Savart law, to show that for Faraday-induced electric fields. (b) Referring to Prob. 5.50a, show that where A is the...
-
A social venture is a business that seeks to solve a social problem and provide a social good. A social good is a product or service that benefits society. Clean air, clean water, literacy, and...
-
Presented here are the accounts of Golden City Barbershop for the year ended December 31, 2016. Land.......... $ 5,000 Notes Payable....... 37,000 Property Tax Expense.... 2,700 Dividends...........
-
1 Define Management? Explain its features, importance and nature? 2 Briefly explain about functions of management 3 Define leadership and explain leadership styles? 4 Briefly explain about decision...
-
The proposed rates were not in the range the CEO expected given the pricing analysis. The CEO has asked the pricing actuary to verify the total projected loss cost excluding potential large storm...
-
10. On which among the following, the speed of the sound depends? 1. Amplitude 2. Wavelength 3. Humidity Select the correct option from the codes given below: [A] Only 3 [B] Only 2 & 3 [C] 1, 2 & 3...
-
A flywheel slows from 484 to 400 rev/min while rotating through 42 revolutions. a. What is the (constant) angular acceleration of the flywheel? rad/s b. How much time elapses during the 42...
-
You require a return of 11.0% on Bulldog stock. You expect next year's EPS (EPS1) to be $9.12 and a dividend payout ratio of 0.6. Bulldog will reinvest the remainder of earnings in projects with...
-
1.Why is capital a more important measure of the size of a securities firm than the amount of assets? 2. What other measures would be useful given the diversity of this industry?
-
Assuming a short-term profit-oriented perspective, the firm would prefer to produce one of the products more than the other. Given this perspective, which product would the firm prefer? Goransson...
-
Why do you think OOFOS s return on ad spend ( ROAS ) might be decreasing over time?
-
Draw the shear, axial, and moment diagrams for the frame below. Draw the free body diagrams, show cuts, and show the shear, axial, and moment functions. What are the reactions at the boundaries? Show...
-
Evenflow Power Co. is considering a new project that is a little riskier than the current operations of the company. Thus, management has decided to add an additional 1.5% to the company's overall...
-
Carbon and silicon belong to Group 4A of the periodic table and have the same valence electron configuration (ns2np2). Why does silicon dioxide (SiO2) have a much higher melting point than carbon...
-
What is the original molarity of a solution of formic acid (HCOOH) whose pH is 3.26 at equilibrium?
-
Metallic iron crystallizes in a cubic lattice. The unit cell edge length is 287 pm. The density of iron is 7.87 g/cm3. How many iron atoms are within a unit cell?
-
Depository institutions have been losing their advantage over other financial intermediaries in attracting customers funds. Why?
-
In the aftermath of the financial crisis of 20072009, there were calls to reinstate the separation of commercial and investment banking activities that was removed with the repeal of the...
-
The globalization of banking has led to the need for global benchmarks for interest rates. In light of the LIBOR scandal, what characteristic do you think is most central to any new interest rate...

Study smarter with the SolutionInn App



