Question: Write a Lewis structure for ethyl alcohol, CH,CH,OH, showing all valence electrons. You do not have to consider stereochemistry. Explicitly draw all H atoms.

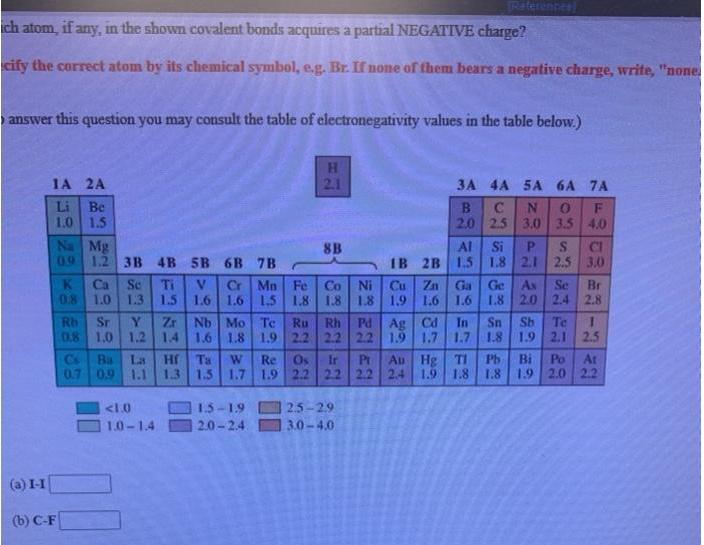


Write a Lewis structure for ethyl alcohol, CH,CH,OH, showing all valence electrons. You do not have to consider stereochemistry. Explicitly draw all H atoms. Include all valence lone pairs in your answer. Raterences ich atom, if any, in the shown covalent bonds acquires a partial NEGATIVE charge? cify the correct atom by its chemical symbol, e.g. Br. If none of them bears a negative charge, write, "none answer this question you may consult the table of electronegativity values in the table below.) 1A 2A 2.1 3A 4A 5A 6A 7A Be 1.0 1.5 Na Mg 09 12 3B 4B 5B 6B 7B Li 2.0 2.5 3,0 3.5 4.0 Al Si P. IB 2B 1.5 1.8 2.1 2,5 3.0 8B CI Cr Mn Fe 1.6 1.6 1.5 1.8 1.8 Co Ni Cu Zn 1.8 1.9 1.6 Ga Ge K Ca 0.8 Se Ti 1.3 Se Br 2.4 2.8 As 1.0 1.5 1.6 1.8 2.0 Zr 08 1.0 1.2 14 Nb Mo Te Pd Ag Cd 1.9 Rh Te Sb 1.9 2.1 2.5 Sr Rh In Sn 1.7 1.7 1.8 Ru 1.6 1.8 1.9 2.2 2.2 2.2 Au Hg TI Cs Bs 0.7 09 1.1 La Hr Ta 1.3 1.5 1.7 1.9 2.2 22 2.2 2.4 Ir Pr Pb B 1.8 Po At 1.9 2.0 2.2 W Re Os 1.9 1.8
Step by Step Solution
There are 3 Steps involved in it
1 Lewis ... View full answer

Get step-by-step solutions from verified subject matter experts


