Write an EES Procedure to calculate COP and WbyQ for a Thermodynamically Ideal, Isobaric source system,...
Fantastic news! We've Found the answer you've been seeking!
Question:
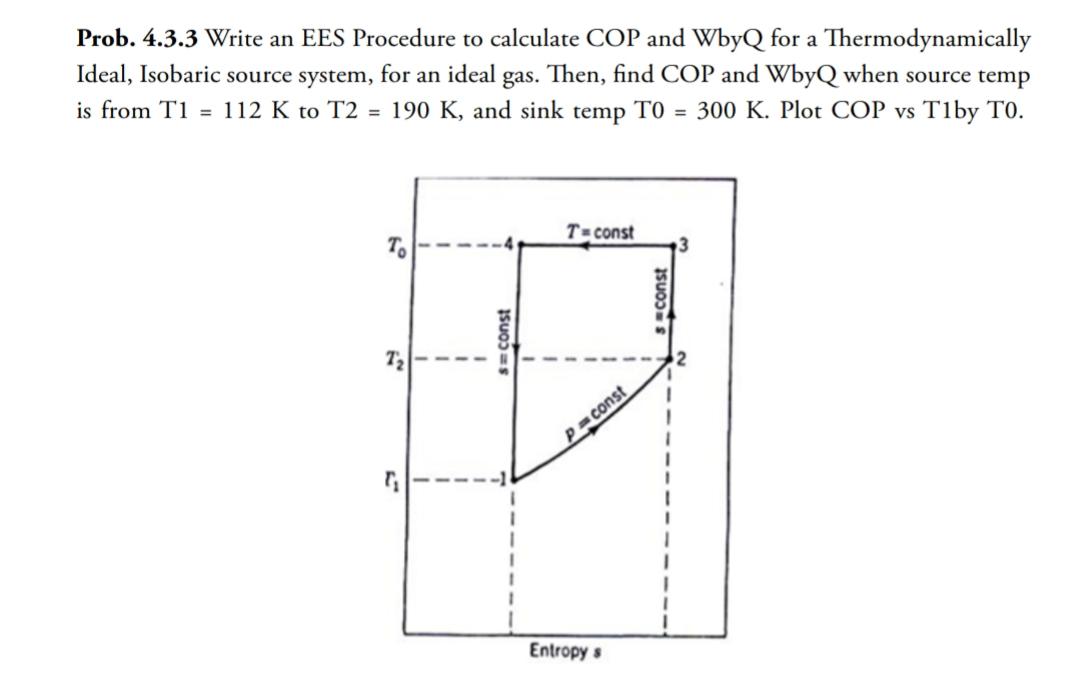
Transcribed Image Text:
Write an EES Procedure to calculate COP and WbyQ for a Thermodynamically Ideal, Isobaric source system, for an ideal gas. Then, find COP and WbyQ when source temp is from T1 112 K to T2 = 190 K, and sink temp TO = 300 K. Plot COP vs T1by TO. To E FM9 I =const T=const P=const Entropy s sconst Write an EES Procedure to calculate COP and WbyQ for a Thermodynamically Ideal, Isobaric source system, for an ideal gas. Then, find COP and WbyQ when source temp is from T1 112 K to T2 = 190 K, and sink temp TO = 300 K. Plot COP vs T1by TO. To E FM9 I =const T=const P=const Entropy s sconst
Expert Answer:
Related Book For 

Introduction to Chemical Engineering Thermodynamics
ISBN: 978-0071247085
7th edition
Authors: J. M. Smith, H. C. Van Ness, M. M. Abbott
Posted Date:
Students also viewed these mechanical engineering questions
-
Write an EES Procedure to calculate the COP and Work requirement for a Thermodynamically Ideal, Isothermal source system.[1]. And, plot the COP against various source temps. Tc for a constant sink...
-
An ideal gas undergoes an isobaric compression and then an isovolumetric process that brings it back to its initial temperature. Had the gas undergone one isothermal process instead, (a) The work...
-
An ideal gas initially at 300 K undergoes an isobaric expansion at 2.50 kPa. If the volume increases from 1.00 m3 to 3.00 m3 and 12.5 kJ is transferred to the gas by heat, what are (a) the change in...
-
1. Analyze whether Thomas can recover his mReport You Need To Prepare: You must prepare a report addressed to Winnie and Ralph. Winnie and Ralph will use the report in discussion with the Clean...
-
Fitzpatrick Inc. planned and manufactured 500,000 units of its single product in 2010, its first year of operations. Variable manufacturing cost were $40 per unit of production. Planned fixed...
-
What are the 17 principles of internal control defined by COSO?
-
Two parallel-plate capacitors have the same plate area. Capacitor 1 has a plate separation twice that of capacitor 2 , and the quantity of charge you place on capacitor 1 is twice the quantity you...
-
Amana Cement Corporation is a private corporation controlled by Amin Amana. The company's adjusted trial balance and other related data at 31 December 20X5 are given below. Although the company uses...
-
QUESTION TWO a) State and explain five monetary policy instruments used by the central bank to regulate interest rate, inflation rate, exchange rate and the quantity of money in the economy [15...
-
TLM Corporations accounting records include the following items, listed in no particular order, at December 31, 2025: The income tax rate for TLM Corporation is 21%. Prepare TLM Corporations income...
-
Use SQL for the following 1. Create table CUSTOMER with following details; 2. Insert rows shown in Fig. 1. 2. Add a column called "Gender" in CUSTOMER 3. Add a column called "Email" and another...
-
Consider an economy characterized by the following facts: i. The official budget deficit is \(4 \%\) of GDP. ii. The debt-to-GDP ratio is \(100 \%\). iii. The nominal interest rate is \(10 \%\). iv....
-
Expansion plc has consistently expanded its activities over the past decade, partly through takeover and partly through organic growth using retention of much of its annual profits. Consolidation plc...
-
More recently, Fama and French (2020) proposed a modified version of their five-factor model in which the multifactors are replaced by long/short mimicking portfolios estimated from a cross-sectional...
-
Write the Fama and French (2018) six-factor model. They expanded this model to include different variations of these factors for a total of 48 factors. Which model worked the best?
-
Lettau and Pelger (2020) proposed the use of Principal Component Analysis (PCA) to identify latent asset pricing factors. How does PCA find factors in stock returns? How many latent factors did they...
-
Let's say you purchase an AAA-rated, 10% coupon, 20-year maturity, $1000 par value corporate bond today for $1085. You hold it for 5 years, re-invest the semi-annual coupons at 5% per year and sell...
-
Use the T account for Cash below to record the portion of each of the following transactions, if any that affect cash. How do these transactions affect the companys liquidity? Jan. 2 Provided...
-
Wet steam at 1,100 kPa expands at constant enthalpy (as in a throttling process) to 101.33 kPa, where its temperature is 105oC. What is the quality' of the steam in its initial state?
-
A Carnot engine is coupled to a Carnot refrigerator so that all of the work produced by the engine is used by the refrigerator in extraction of beat from a heat reservoir at 0C at the rate of 35 kJ...
-
Calculate Z and V for ethylene at 25(C and 12 bar by the following equations: (a) The truncated virial equation [Eq. (3.401] with the following experimental values of virial coefficients: (b) The...
-
Determine the equivalent resistance \(R_{\text {eq }}\) for the circuit shown in Figure 6.8. FIGURE 6.8 Problem 1. W R1 ev
-
Determine the equivalent resistance \(R_{\text {eq }}\) for the circuit shown in Figure 6.9. FIGURE 6.9 Problem 2. +O V www R ww R3
-
Determine the equivalent resistance \(R_{\text {eq }}\) for the circuit shown in Figure 6.10. Assume that all resistors have the same resistance of \(R\). FIGURE 6.10 Problem 3. W R . WWW R5 w R2 R3...

Study smarter with the SolutionInn App



