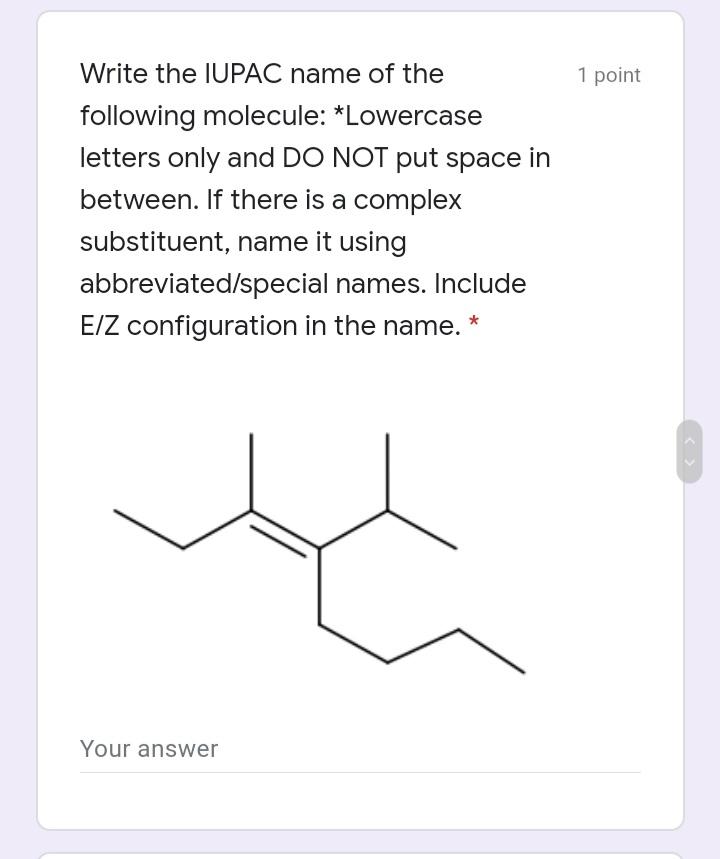
Write the IUPAC name of the 1 point following molecule: *Lowercase letters only and DO NOT...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
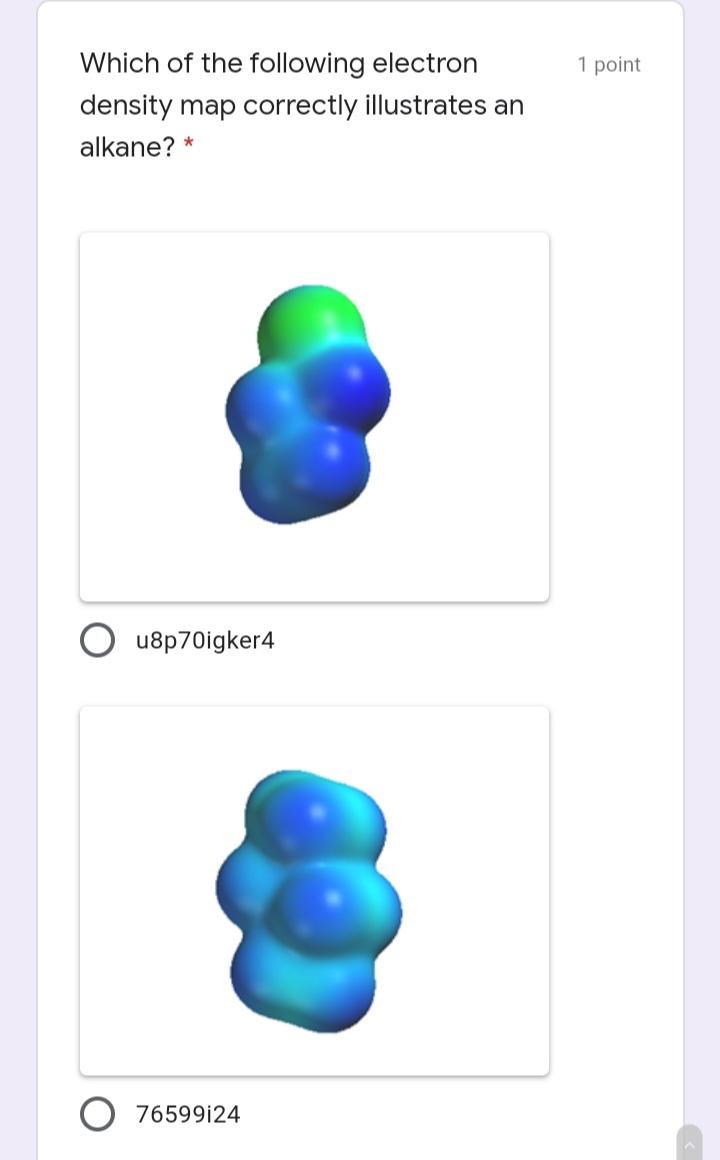
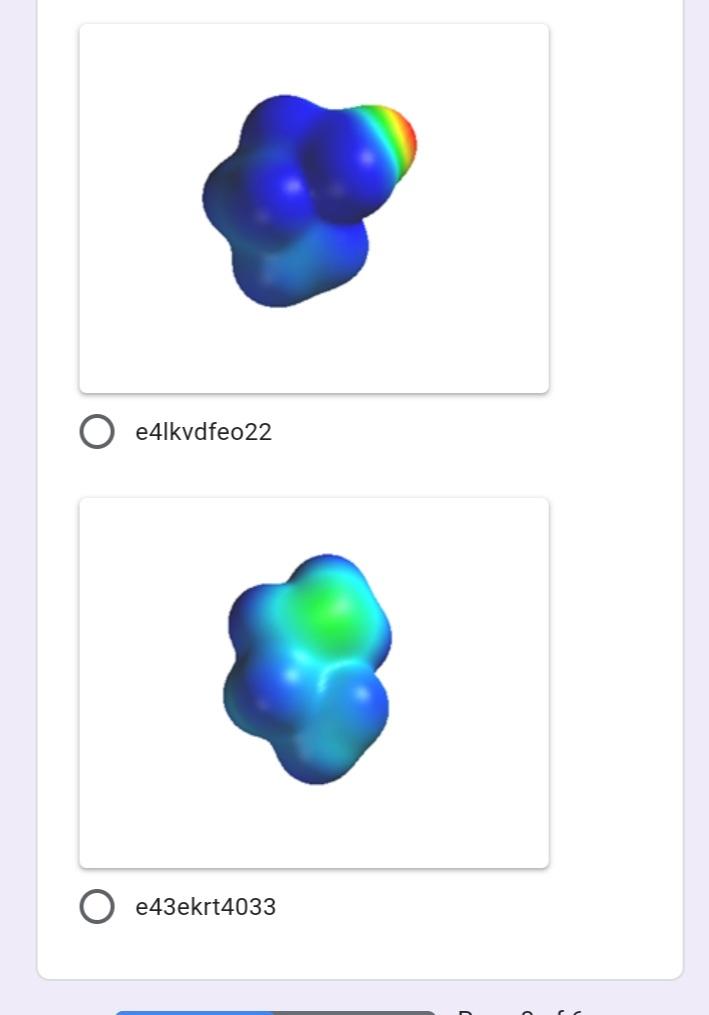
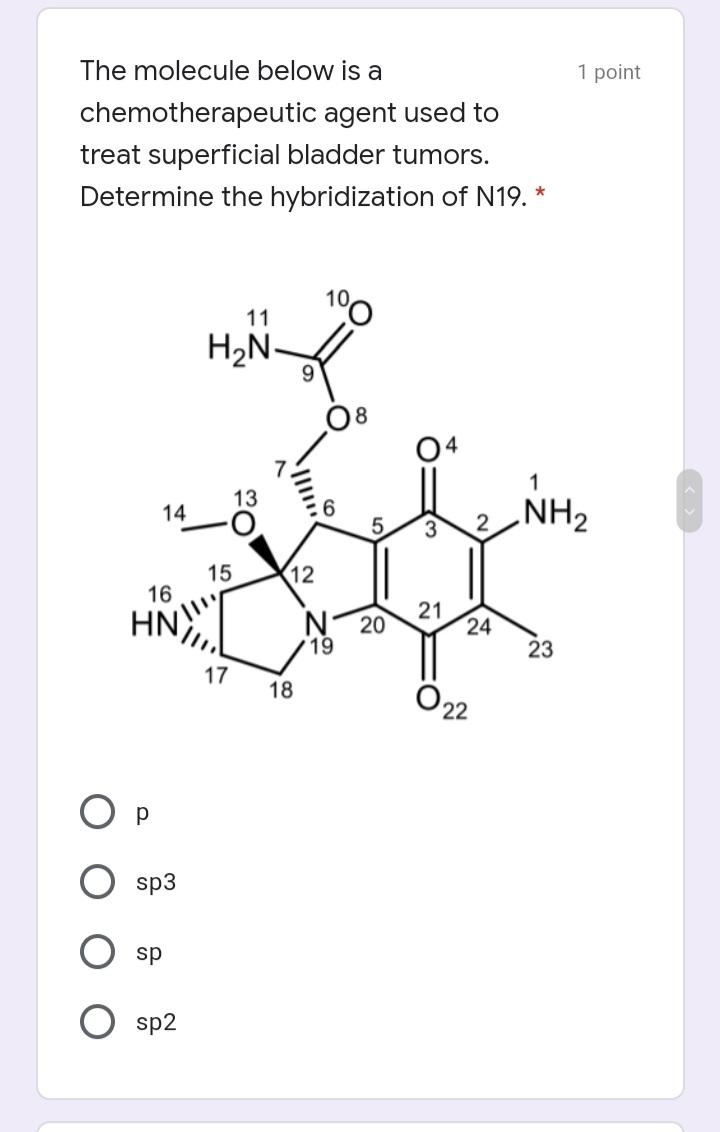
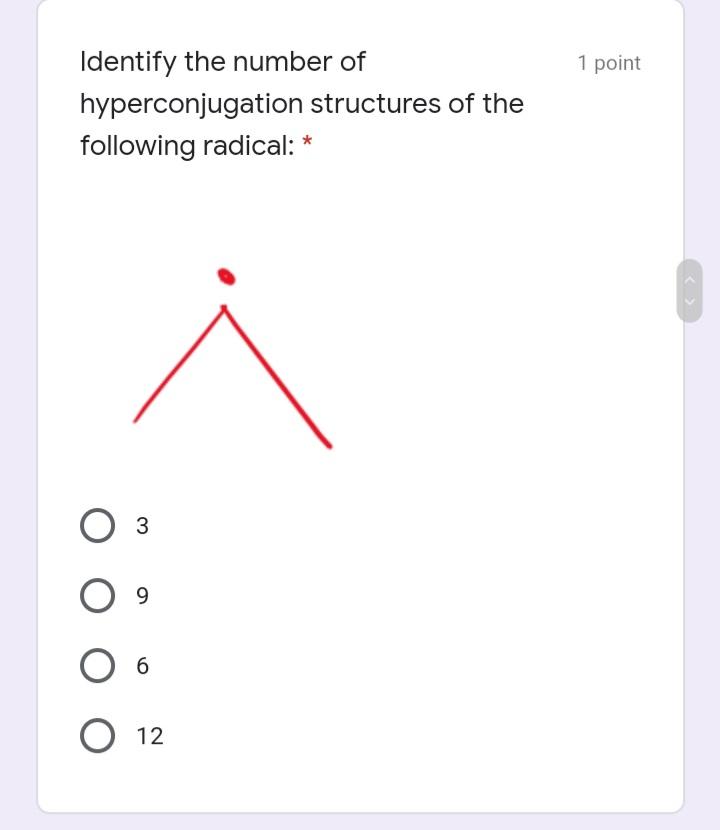
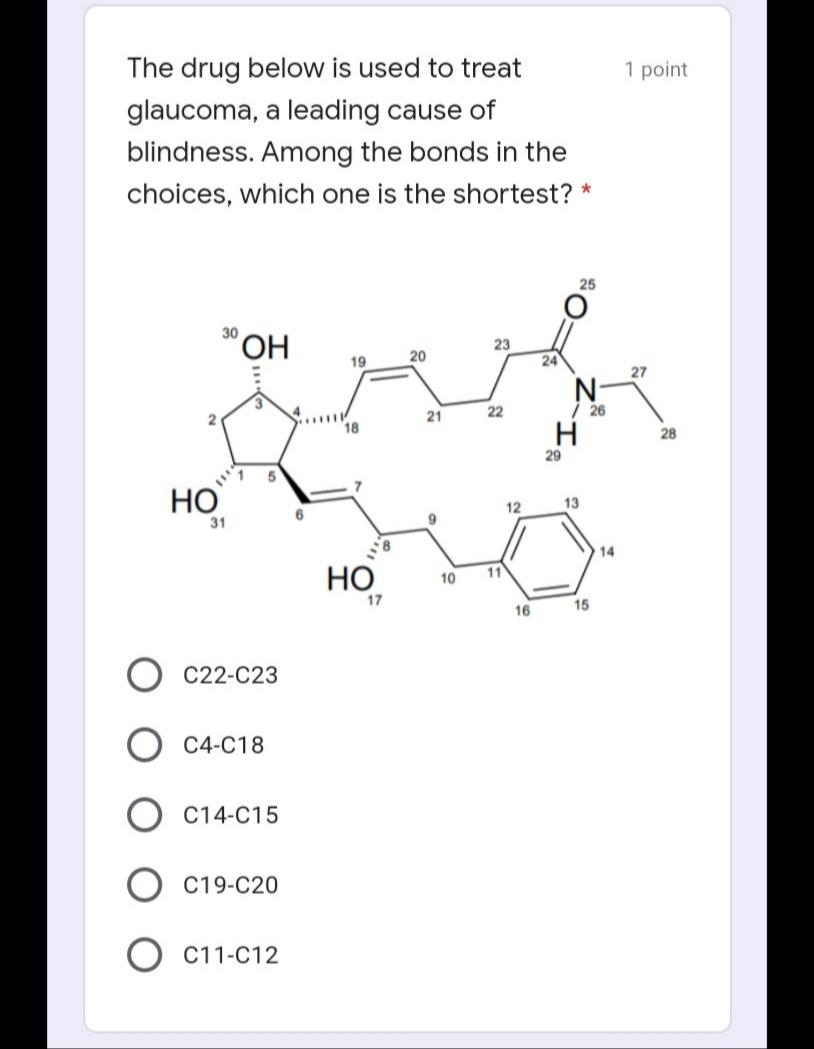
Write the IUPAC name of the 1 point following molecule: *Lowercase letters only and DO NOT put space in between. If there is a complex substituent, name it using abbreviated/special names. Include E/Z configuration in the name. * Your answer Which of the following electron 1 point density map correctly illustrates an alkane? * u8p70igker4 O 76599i24 e4lkvdfeo22 e43ekrt4033 The molecule below is a 1 point chemotherapeutic agent used to treat superficial bladder tumors. Determine the hybridization of N19. * 11 H2N- 6. 04 13 14 2 NH2 15 12 16 N 20 19 21 24 23 17 18 22 sp3 sp sp2 Identify the number of 1 point hyperconjugation structures of the following radical: 3 9. 6. 12 The drug below is used to treat 1 point glaucoma, a leading cause of blindness. Among the bonds in the choices, which one is the shortest? * 25 30 HO, 23 19 20 24 27 22 / 26 21 H. 18 28 29 5 НО 12 13 31 14 НО 11 10 17 15 16 C22-C23 C4-C18 C14-C15 O C19-C20 O C11-C12 Write the IUPAC name of the 1 point following molecule: *Lowercase letters only and DO NOT put space in between. If there is a complex substituent, name it using abbreviated/special names. Include E/Z configuration in the name. * Your answer Which of the following electron 1 point density map correctly illustrates an alkane? * u8p70igker4 O 76599i24 e4lkvdfeo22 e43ekrt4033 The molecule below is a 1 point chemotherapeutic agent used to treat superficial bladder tumors. Determine the hybridization of N19. * 11 H2N- 6. 04 13 14 2 NH2 15 12 16 N 20 19 21 24 23 17 18 22 sp3 sp sp2 Identify the number of 1 point hyperconjugation structures of the following radical: 3 9. 6. 12 The drug below is used to treat 1 point glaucoma, a leading cause of blindness. Among the bonds in the choices, which one is the shortest? * 25 30 HO, 23 19 20 24 27 22 / 26 21 H. 18 28 29 5 НО 12 13 31 14 НО 11 10 17 15 16 C22-C23 C4-C18 C14-C15 O C19-C20 O C11-C12
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
An economics student wishes to see if there is a relationship between the amount of state debt per capita and the amount of tax per capita at the state level. Based on the following data, can she or...
-
A study was conducted to determine if there is a difference between the investing preferences of mid-level managers working in the public and private sectors in New York City. A random sample of 320...
-
Write the IUPAC Name of the following structure
-
Rainfall of magnitude 3.8cm and 2.8cm occurring on two consecutive 4-h durations on a catchment of area 27km produced the following hydrograph of flow at the outlet of the catchment. Estimate the...
-
Find the depreciation each year for a tractor that was purchased for $18,000 and placed in service midyear under the MACRS method of depreciation as a 3-year property.
-
What are the advantages to including a cutoff score as a means of assignment in the regression-discontinuity approach?
-
For a specially orthotropic, transversely isotropic material the "plane strain bulk modulus," \(K_{23}\), is an engineering constant that is defined by the stress conditions...
-
You are the in charge auditor examining the financial statements of the Gutzler Company for the year ended December 31. During late October, with the help of Gutzlers controller, you completed an...
-
HR Competencies & Applications Setting up an Alternative Dispute Resolution Process Creating an internal process to settle employment-related disputes and conflicts can benefit both the organization...
-
It is January 1, 2017 and Pegasus is contemplating the acquisition of competitor Chimera. The following details are available (S in millions except per share data): January 1, 2017 (S in millions)...
-
A security camera is 10 ft from the center of a desk that 10 feet long. What angle should the camera move so that it gets a view of the entire desk?
-
9 101) Cakulate EP unit costs per ingredient. Next find total value of the inventory on hand for the end of the month of June. 11 2) Calculate cost of goods sold (sales) 12 31 Cost out each...
-
A hot - air balloon is descending at a rate of 2 . 4 m / s when a passenger drops a camera. If the camera is 4 2 m above the ground when it is dropped, what is its velocity just before it lands? Let...
-
Translate the following code into MIPS code. int Test (int i, int j) { int a; a = i + j + 5; a = Leaf (a + 1) + Leaf (a); return a; } int Leaf (int b) { int c; c = b+b; return g; } Assume variables a...
-
Regarding your social issue, describe your realistic personal action plan for moving from your current stage of the Cycle of Liberation to the next stage in the Cycle by creating three SMART goals...
-
A loudspeaker has a power output of 2 6 0 watts, calculate the sound intensity in watt / meter ^ 2 at a distance of 5 meter from the loudspeaker, if the power is spread over half the surface area of...
-
Assume that the spot-rate curve is flat at 6%. Consider a portfolio consisting of two par-coupon bonds. (Recall that if the spot-rate curve is flat, then the par-coupon yield curve is also flat at...
-
2. In the circuit given in Figure 2, i,(t) = 5.67cos(5t)A and v (t) = 70.71 cos(5t 60) V a) Find the equivalent load impedance. State whether the load is inductive or capacitive. b) Calculate the...
-
For each of the following statements, indicate whether it is true or false. a. A spontaneous reaction always releases heat. b. A spontaneous reaction is always a fast reaction. c. The entropy of a...
-
The ionization energy of O2 is smaller than the ionization energy of atomic O; the opposite is true for the ionization energies of N2 and atomic N. Explain this behavior in terms of the molecular...
-
Describe the major trends that emerge when atomic radii are plotted against atomic number. Describe the trends observed when first ionization energies are plotted against atomic number.
-
Let \(A\) and \(B\) be disjoint events with \(P(A)=0.3\) and \(P(B)=0.45\). Determine the probabilities \(P(A \cup B), P(\overline{A \cup B}), P(\bar{A} \cup \bar{B})\), and \(P(\bar{A} \cap B)\).
-
Let \(P(A \cap \bar{B})=0.3\) and \(P(\bar{B})=0.6\). Determine \(P(A \cup B)\).
-
Check whether for 3 arbitrary random events \(A, B\), and \(C\) the following constellations of probabilities can be true: (1) \(P(A)=0.6, P(A \cap B)=0.2\), and \(P(A \cap \bar{B})=0.5\), (2)...

Study smarter with the SolutionInn App


