Write the k & kp equation of the followings: I. 2NO2N2O4 . H2 + I2 ...
Fantastic news! We've Found the answer you've been seeking!
Question:
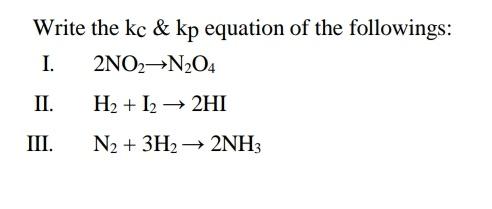
Transcribed Image Text:
Write the kç & kp equation of the followings: I. 2NO2→N2O4 П. H2 + I2 → 2HI III. N2 + 3H2 → 2NH3 Write the kç & kp equation of the followings: I. 2NO2→N2O4 П. H2 + I2 → 2HI III. N2 + 3H2 → 2NH3
Expert Answer:
Answer rating: 100% (QA)
Kc and Kp are e eguilitorium Constem ts 4 gasea mixture REI... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The following reaction is at equilibrium: N2 + 3H2 + 2NH3 The Keq at a particular temperature is 13.7. If the equilibrium [N2] is 0.055 M and the equilibrium [H2] is 1.62 M, what is the equilibrium...
-
Write expressions for K and Kp for the following reactions. a. 2NH3(g) + CO2(g) N2CH4O(s) + H2O(g) b. 2NBr3(s) N2(g) + 3Br2(g) c. 2KClO3(s) 2KCl(s) + 3O2(g) d. CuO(s) + H2(g) Cu(l) + H2O(g)
-
The decomposition of NH3 to N2 and H2 was studied on two surfaces: Without a catalyst, the activation energy is 335 kJ/ mol. a. Which surface is the better heterogeneous catalyst for the...
-
47GP: Chapter: CH0 CH1 CH2 CH3 CH4 CH5 CH6 CH7 CH8 CH9 CH10 CH11 CH12 CH13 CH14 CH15 CH16 CH17 CH18 CH19 CH20 CH21 CH22 CH23 CH24 CH25 CH26 CH27 CH28 CH29 CH30 Problem: 1CQ 1MCP 1P 2CQ 2MCP 2P 3CQ...
-
On December 31, 2011, IBM Corporation had 4,687.5 million shares of common stock authorized. There were 2,182.5 million shares issued, and 1,019.3 million shares held as treasury stock. How many...
-
The College Athletes Players Association petitioned the National Labor Relations Board (NLRB) contending that football players at Northwestern University who receive "grant-in-aid" scholarships are...
-
A centrifugal pump having a 6-in.-diameter impeller and the characteristics shown in Fig. 12.7 is to be used to pump gasoline through \(4000 \mathrm{ft}\) of commercial steel 3-in.-diameter pipe. The...
-
The following transactions apply to Walnut Enterprises for 2016, its first year of operations: 1. Received $50,000 cash from the issue of a short-term note with a 6 percent interest rate and a...
-
Explain how RTOS are utilized in embedded systems for industrial, automotive, or aerospace applications. What are the key design principles for integrating RTOS into embedded devices with strict...
-
Jenny Smith, 28, just received a promotion at work. Her salary has increased to $40 000 and she is now eligible to participate in her employers pension plan. The employer matches employee...
-
g) Being able to use a computer is inversely related to being depressed. Does this imply that educating elderly about computer usage would reduce depression among this group?
-
Write a program that takes the name of a .wav file and a playback rate r as command-line arguments and plays the file at the given rate. First, use StdAudio.read() to read the file into an array a[]....
-
A simple pendulum, with mass \(m=60 \mathrm{~g}\), and inextensible wire \(L=120 \mathrm{~cm}\) long, starts from rest initially at an angle of \(60^{\circ}\) to the vertical. Determine the speed...
-
Indicate true or false for the following statements. If false, specify what change will make the statement true. _______________________ If every observation is multiplied by 2, then the t statistic...
-
Which of the following is the correct order of phases of the eukaryotic cell cycle? a. G1, G2, S, M b. G1, S, G2, M c. G1, G2, M, S d. G1, S, M, G2
-
This section consists of some true/false questions regarding concepts of statistical inference. Indicate if a statement is true or false and, if false, indicate what is required to make the statement...
-
P 1 1 Describe two iterative and two sequential software lifecycle models. P 2 2 Explain how risk is managed in software lifecycle models.
-
A regular deposit of $100 is made at the beginning of each year for 20 years. Simple interest is calculated at i% per year for the 20 years. At the end of the 20-year period, the total interest in...
-
What is a disproportionation reaction? Use the following reduction potentials ClO3- + 3H+ + 2e- HClO2 + H2O o = 1.21 V HClO2 + 2H+ + 2e- HClO + H2O o = 1.65 V To predict whether HClO2 will...
-
The unit cell of MgO is shown below. Does MgO have a structure like that of NaCl or ZnS? If the density of MgO is 3.58 g/cm3, estimate the radius (in centimeters) of the O2- anions and the Mg2+...
-
Sorbic acid is used to prevent mold and fungus growth in some food products, especially cheeses. The systematic name for sorbic acid is 2,4-hexadienoic acid. Draw structures for the four geometric...
-
The PFD in Figure 11.63 shows a process in which two liquid products, A and B, are produced from a feed stream of raw material R. In the process, the reactor feed is preheated to \(300^{\circ}...
-
Consider a process with the following streams: (a) Compute \(\Delta T_{\text {thres }}\) as well as the minimum external heating and cooling requirements as a function of \(\Delta T_{\text {min }}\)....
-
Design a HEN to meet the MER targets for \(\Delta T_{\text {min }}=10^{\circ} \mathrm{C}\) and \(N_{H X, \text { min }}\) for a process involving five hot streams and one cold stream as introduced by...

Study smarter with the SolutionInn App


