Write the molecular and empirical formulas of the following compounds: H H (a) (b) H (c)...
Fantastic news! We've Found the answer you've been seeking!
Question:
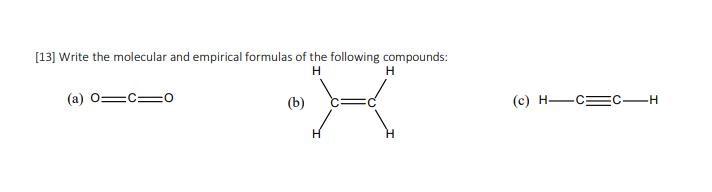
Transcribed Image Text:
Write the molecular and empirical formulas of the following compounds: H H (a) (b) H (c) H-CE C-H Write the molecular and empirical formulas of the following compounds: H H (a) (b) H (c) H-CE C-H
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Write the molecular and structural formulas for the compounds represented by the following molecular models: (a) (b) (c) (d) P F F F
-
Write the molecular and structural formulas for the compounds represented by the following models: (a) (b) (c) (d) Cl Cl
-
Determine the molecular and empirical formulas of the compounds shown here. (Black spheres are carbon and gray spheres are hydrogen.) (b) (c) (d) (a)
-
Built-Tite uses job order costing. The T-account below summarizes Factory overhead activity for the current year. Factory Overhead Debit Credit 16,200 106,600 25,200 60,200 1. Compute total applied...
-
The Blair Companys three assembly plants are located in California, Georgia, and New Jersey. Previously, the company purchased a major subassembly, which becomes part of the final product, from an...
-
At the end of 2019, Lucretia McEvil Company has $180,000 of cumulative temporary differences that will result in reporting the following future taxable amounts. 2020...........................$...
-
Describe the function of each branch of government.
-
Mayfield, Inc., has the following plant asset accounts: Land, Buildings, and Equipment, with a separate accumulated depreciation account for each of these except Land. Mayfield completed the...
-
Does the game have a dominant-strategy equilibrium? If so, what is it and why is it that? If not, why not?
-
Robson Electronics completed these selected transactions during March 2018: a. Sales of $2,050,000 are subject to an accrued warranty cost of 9%. The accrued warranty payable at the beginning of the...
-
A) Identify Atoms That Have Charge From Equation below. B) By Showing The Charged (If Present), Rewrite Each Of The Above Reactions And Show The Curved Arrows (Mechanism) CH3OH + HI CH3OH2+ + I-...
-
We are the UKs leading home and general merchandise retailer. Argos and Homebase are two of the UKs leading retail brands, with large customer bases across the UK and Ireland. Between them, our...
-
The House of Commons Committee of Public Accounts produced a report on the budget for the London 2012 Olympic and Paralympic Games, to investigate the reasons for budget increases announced by the...
-
(a) Using the information provided in question C12.1, prepare a spreadsheet containing a trial balance, adjustment and resulting figures for income statement (profit and loss account) and statement...
-
In the following extract, the College of Charleston, USA, describes on its website how it estimates the standard cost of attendance in respect of a student. To consider a student for federal...
-
Prepare ledger accounts to report the transactions and events of questions C8.1 and C8.2. Data from C8.1 The Biscuit Manufacturing Company commenced business on 1 January Year 1 with capital of...
-
Formal and informal institutions are important in shaping business strategies for specific countries and markets. Answer the following: a) Describe, using examples, the major political, economic and...
-
In a large midwestern university, 30% of the students live in apartments. If 200 students are randomly selected, find the probability that the number of them living in apartments will be between 55...
-
Calculate the mass in grams of hydrogen chloride produced when 5.6 L of molecular hydrogen measured at STP react with an excess of molecular chlorine gas.
-
The equilibrium constant KP for the reaction is 5.60 Ã 104 at 350°C. The initial pressures of SO2 and O2 in a mixture are 0.350 atm and 0.762 atm, respectively, at 350°C. When the...
-
What masses of sodium chloride, magnesium chloride, sodium sulfate, calcium chloride, potassium chloride, and sodium bicarbonate are needed to produce 1 L of artificial seawater for an aquarium? The...
-
In 2022, Mark purchased two separate activities. Information regarding these activities for 2022 and 2023 is as follows: The 2022 losses were suspended losses for that year. During 2023, Mark also...
-
Jerry sprayed all of the landscaping around his business with a pesticide in June 2023. Shortly thereafter, all of the trees and shrubs unaccountably died. The FMV and the adjusted basis of the...
-
In 2023, Julie, a single individual, reported the following items of income and deduction: Julie owns 100% and is an active participant in the rental real estate activity. What is her taxable income...

Study smarter with the SolutionInn App



