You are a biochemist studying the lactase enzyme, and you are purifying the enzyme from a bacterial
Question:
You are a biochemist studying the lactase enzyme, and you are purifying the enzyme from a bacterial lysate. After an initial ammonium sulfate precipitation step followed by dialysis of your sample, you are optimizing a chromatography using a stationary phase composed of diethylaminoethyl cellulose (which contains ionizable amino groups) and a mobile phase initially composed of 10mM phosphate buffer pH 8. You apply your sample and collect 10 1 mL fractions, then proceed to change the mobile phase so that the NaCl concentration in the buffer gradually increases from 0 to 0.5M NaCl, collecting 40 more 1 mL fractions during this gradient. For each fraction, you?
determine?
1) the presence of protein by absorbance and?
2) the lactase enzymatic activity.
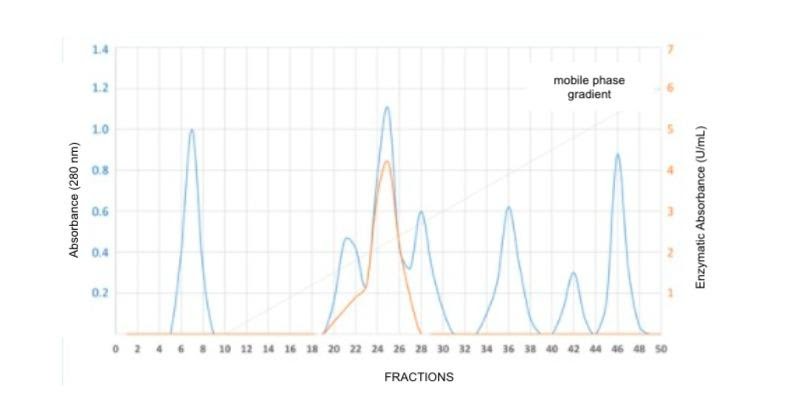
Based on this information, what is the charge of the proteins eluted in fractions 5-9 and what would be a reasonable value for the pI of lactase??
?




