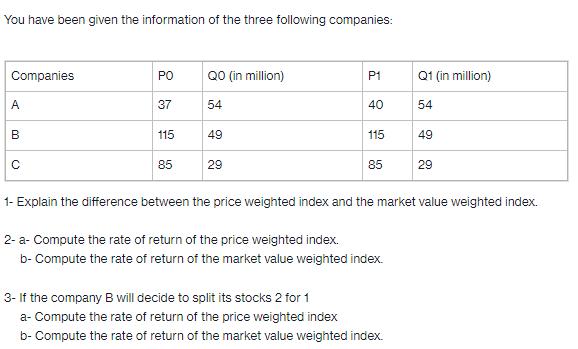
You have been given the information of the three following companies: Companies A B PO...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
You have been given the information of the three following companies: Companies A B с PO 37 115 85 QO (in million) 54 49 29 P1 40 115 85 2- a- Compute the rate of return of the price weighted index. b- Compute the rate of return of the market value weighted index. Q1 (in million) 54 3- If the company B will decide to split its stocks 2 for 1 a- Compute the rate of return of the price weighted index b- Compute the rate of return of the market value weighted index. 49 1- Explain the difference between the price weighted index and the market value weighted index. 29 You have been given the information of the three following companies: Companies A B с PO 37 115 85 QO (in million) 54 49 29 P1 40 115 85 2- a- Compute the rate of return of the price weighted index. b- Compute the rate of return of the market value weighted index. Q1 (in million) 54 3- If the company B will decide to split its stocks 2 for 1 a- Compute the rate of return of the price weighted index b- Compute the rate of return of the market value weighted index. 49 1- Explain the difference between the price weighted index and the market value weighted index. 29
Expert Answer:
Answer rating: 100% (QA)
1 Difference between Price Weighted Index and Market Value Weighted Index Price Weighted Index In a price weighted index the components of the index a... View the full answer

Related Book For 

Income Tax Fundamentals 2013
ISBN: 9781285586618
31st Edition
Authors: Gerald E. Whittenburg, Martha Altus Buller, Steven L Gill
Posted Date:
Students also viewed these finance questions
-
You have been given the following information on a project: It has a five-year lifetime The initial investment in the project will be $25 million, and the investment will be depreciated straight...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
You are the audit senior who has recently been assigned to the year-end audit of Ontario Agra Corporation (OAC). After the end of the audit fieldwork, the senior on the engagement resigned suddenly...
-
At t = 0, a particle is traveling parallel to very large insulating plate at a 0.360 m distance with a speed of 980 m/s. Consider that the insulating plate has uniform surface charge density 2.34. x...
-
Distinguish between the two primary viewpoints concerning the preparation of interim financial statements.
-
From a heat transfer point of view, what is the difference between isotropic and anisotropic materials?
-
1. To develop an understanding of your ethical leadership style 2. To understand how your preferred ethical leadership style relates to other ethical leadership styles Directions 1. Please read the...
-
Spamela Hamderson Inc. reports the following pretax income (loss) for both financial reporting purposes and tax purposes. (Assume the carryback provision is used for a net operating loss.) The tax...
-
Based on the above case study, "Want-2-Work", present each of the following: 1) ER diagram 2) Relational model 3) Rules or assumptions made 4) Between 3 or 5 queries using the relational model...
-
Consider the hypothetical eutectic phase diagram for metals A and B, which is similar to that for the lead-tin system, Figure 9.8. Assume that (1) and phases exist at the A and B extremities of the...
-
Let Xn and Yn be independent random variables for all n such that Xn+Yn 0. Prove that there exists a sequence {an}1 CR such that Xn - an , 0. (Hint: consider the medians.)
-
According to an autograph association, only 6% of autographs in circulation from a certain band are estimated to be real. If there are 74 authentic autographs of the band in circulation, then how...
-
The Apollo's Chariot is a rollercoaster at Busch Gardens with a top speed of 117 km/hr. Its initial (and tallest) descent can be modeled by h(t) = 52 - 4.912, where his in meters and i is in seconds....
-
Minimize the following Boolean functions using K-maps: a) f (a, b, c) = (2, 3, 6, 7) = (2, 3, 6, 7, 12, 13, 14) b) f (a, b, c, d) c) f (a, b, c) = (0, 3, 4, 7) d) f (a, b, c, d) = e) f (a, b, c, d)...
-
1) Add the Value 2 to the tree and Balance the AVL Tree 3 5 4 6
-
What is 20% of 90? Write your answer rounded to the nearest whole number. r30% of 240 is what number? Round your answer to the nearest whole number. r230% of what number is 460? Round your answer to...
-
At a distance of 6.4 cm, a 51-mm-high by 89-mm-long business card fills the screen of a cell phone in camera mode. If the focal length of the camera lens is 4.3 mm, what is the length I of the...
-
Controls can be identified based on their function. The functions are preventive, detective, and corrective. A. True B. False
-
Indicate, in each of the following situations, the number of exemptions the taxpayers are entitled to claim on their 2012 income tax returns. Number of Exemptions a. Donna, a 20-year-old single...
-
Ray and Maria Gomez have been married 3 years. They live at 1610 Quince Ave., McAllen, TX 78701. Ray works for Palm Oil Corporation and Maria works for the City of McAllen. Maria's Social Security...
-
Dr. George E. Beeper is a single taxpayer. He lives at 45 Mountain View Dr., Apt. 321, Spokane, WA 99210. Dr. Beeper's Social Security number is 775-88-9531. Dr. Beeper works for the Pine Medical...
-
How many structural isomers are there for hydrocarbons that have the molecular formula C 4 H 10 ? (a) none (b) one (c) two (d) three
-
Which contains more hydrogen atomsa five-carbon saturated hydrocarbon molecule or a five-carbon unsaturated hydrocarbon molecule? (a) The unsaturated hydrocarbon has more hydrogen atoms. (b) The...
-
Explain why caprylic acid, CH 3 (CH 2 ) 6 COOH, dissolves in a 5, aqueous solution of sodium hydroxide but caprylaldehyde, CH 3 (CH 2 ) 6 CHO, does not dissolve. (a) With two oxygens, the caprylic...

Study smarter with the SolutionInn App


