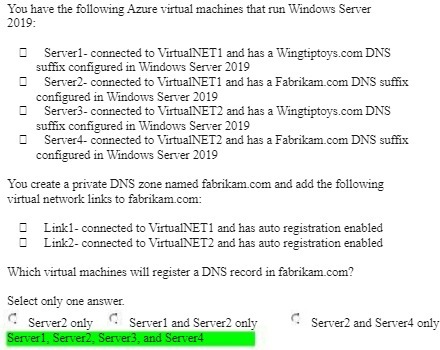
You have the following Azure virtual machines that run Windows Server 2019: Server1-connected to VirtualNET1 and...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
You have the following Azure virtual machines that run Windows Server 2019: Server1-connected to VirtualNET1 and has a Wingtiptoys.com DNS suffix configured in Windows Server 2019 Server2- connected to VirtualNET1 and has a Fabrikam.com DNS suffix configured in Windows Server 2019 Server3-connected to VirtualNET2 and has a Wingtiptoys.com DNS suffix configured in Windows Server 2019 Server4- connected to VirtualNET2 and has a Fabrikam.com DNS suffix configured in Windows Server 2019 You create a private DNS zone named fabrikam.com and add the following virtual network links to fabrikam.com: Link1-connected to VirtualNET1 and has auto registration enabled Link2-connected to VirtualNET2 and has auto registration enabled Which virtual machines will register a DNS record in fabrikam.com? Select only one answer. Server2 only D Server1 and Server2 only Server2 and Server4 only Server1, Server2, Server3, and Server4 You have the following Azure virtual machines that run Windows Server 2019: Server1-connected to VirtualNET1 and has a Wingtiptoys.com DNS suffix configured in Windows Server 2019 Server2- connected to VirtualNET1 and has a Fabrikam.com DNS suffix configured in Windows Server 2019 Server3-connected to VirtualNET2 and has a Wingtiptoys.com DNS suffix configured in Windows Server 2019 Server4- connected to VirtualNET2 and has a Fabrikam.com DNS suffix configured in Windows Server 2019 You create a private DNS zone named fabrikam.com and add the following virtual network links to fabrikam.com: Link1-connected to VirtualNET1 and has auto registration enabled Link2-connected to VirtualNET2 and has auto registration enabled Which virtual machines will register a DNS record in fabrikam.com? Select only one answer. Server2 only D Server1 and Server2 only Server2 and Server4 only Server1, Server2, Server3, and Server4
Expert Answer:

Related Book For 

Cornerstones of Financial and Managerial Accounting
ISBN: 978-1111879044
2nd edition
Authors: Rich, Jeff Jones, Dan Heitger, Maryanne Mowen, Don Hansen
Posted Date:
Students also viewed these algorithms questions
-
Your server must meet the following criteria: Logical naming convention for your company Two NICs (one LAN segment and one NAT) Static IP address, subnet mask and default gateway on the LAN segment...
-
can someone solve this Modern workstations typically have memory systems that incorporate two or three levels of caching. Explain why they are designed like this. [4 marks] In order to investigate...
-
An average of three small businesses go bankrupt each month. What is the probability that five small businesses will go bankrupt in a certain month?
-
Gramps' Remedy manufactures athletes' foot powder. The company uses a standard costing system. Following are data pertaining to the company's operations for 1999: Production for the...
-
The sign is subjected to the uniform wind loading. Determine the stress components at points C and D on the 100-mm-diameter supporting post. Show the results on a volume element located at each of...
-
Calculate an acceptable risk of incorrect acceptance assuming: (1) audit risk is 5 percent, (2) the risk that the internal controls fail to detect material errors is 40 percent, and (3) the risk that...
-
Scandia Coat Company makes women's and men's coats. Both products require filler and lining material. The following planning information has been made available: Scandia Coat does not expect there to...
-
Five years have passed and Jamie Lee. 34, is considering taking the plunge-not only is she engaged to be married, but she is also deciding on whether to purchase a new home. Jamie Lee's cupcake caf...
-
Usonic, Inc., has collected the following information on its cost of electricity: Required a. Prepare a scattergraph of Usonic's electricity costs for the year. Plot the total electricity cost on the...
-
In 2017, Esters Bunny Barn had discontinued and sold its unprofitable manufacturing division. Esters experienced a $200,000 loss from the chemical division. The loss on disposal of the manufacturing...
-
What is the statement of stockholders' equity? What useful information does it contain?
-
Delta Air Lines, Inc. reports total net operating assets of $6,829 million, net nonoperating liabilities of $8,960 million, and equity of $(2.131) in its 2012 10-K. Footnotes reveal the existence of...
-
Explain why using the FIFO inventory costing method will increase gross profit during periods of rising inventory costs.
-
What four different types of adjustments are frequently necessary before financial statements are prepared at the end of an accounting period? Give at least one example of each type.
-
Why is depreciation expense necessary to properly match revenues and expenses?
-
Draw the contributing structure that results from resonance indicated by the curved arrow(s). You do not have to consider stereochemistry. Explicitly draw all H atoms. You do not have to include lone...
-
Provide a draft/outline of legal research involving an indigenous Canadian woman charged with assault causing bodily harm under (Sec 267b) of the Criminal Code, where the crown wants a 12-month jail...
-
Pillsbury Corporation has $65,000 of cost of goods sold and average inventory of $30,000. What is Pillsburys inventory turnover ratio ? a. 0.46 b. 1.17 c. 1.46 d. 2.17
-
Multiple Choice Questions 1. Which of the following use financial statement data to make decisions? a. Customers b. Investors c. Suppliers d. All of these 2. Which statement would best provide...
-
Barb Muller wins the lottery. She wins $ 20,000 per year to be paid for 10 years. The state offers her the choice of a cash settlement now instead of the annual payments for 10 years. Required: If...
-
(a) Describe the effect of temperature on the equilibrium constant. (b) Industrial \(\mathrm{CH}_{3} \mathrm{OH}\) is prepared according to the reaction \[ \mathrm{CO}(\mathrm{g})+2...
-
Under atmospheric conditions the acetone-chloroform azeotrope boils at \(64.6^{\circ} \mathrm{C}\) and contains 33.5 mole per cent acetone. The vapour pressures of acetone and chloroform at this...
-
Explain in detail, with neat sketch, the working principle of a fuel cell in producing electrical energy from the chemical energy of a fuel.

Study smarter with the SolutionInn App


