The figure shows a reversible cycle through which 1.00 mole of a monatomic ideal gas is...
Fantastic news! We've Found the answer you've been seeking!
Question:
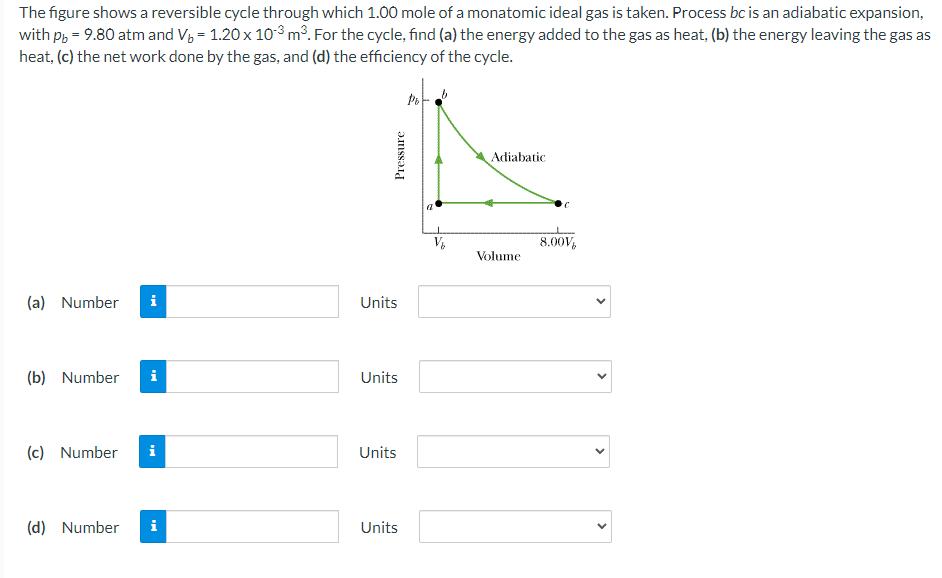
Transcribed Image Text:
The figure shows a reversible cycle through which 1.00 mole of a monatomic ideal gas is taken. Process bc is an adiabatic expansion, with pb = 9.80 atm and V₂ = 1.20 x 103 m³. For the cycle, find (a) the energy added to the gas as heat, (b) the energy leaving the gas as heat, (c) the net work done by the gas, and (d) the efficiency of the cycle. (a) Number i (b) Number Mi (c) Number i (d) Number Mi Pressure Units Units Units Units V₂ Adiabatic Volume 8.00V > > The figure shows a reversible cycle through which 1.00 mole of a monatomic ideal gas is taken. Process bc is an adiabatic expansion, with pb = 9.80 atm and V₂ = 1.20 x 103 m³. For the cycle, find (a) the energy added to the gas as heat, (b) the energy leaving the gas as heat, (c) the net work done by the gas, and (d) the efficiency of the cycle. (a) Number i (b) Number Mi (c) Number i (d) Number Mi Pressure Units Units Units Units V₂ Adiabatic Volume 8.00V > >
Expert Answer:
Answer rating: 100% (QA)
Number of moles n 1 Pressure at b is Pb 15 atm 15 x 101 x10 5 Pa Volume at b is Vb 12 x... View the full answer

Related Book For 

Management information systems
ISBN: 978-0073376813
10th edition
Authors: James A. O Brien, George M. Marakas
Posted Date:
Students also viewed these physics questions
-
Assume that in your first week on a new job you are asked to use a type of business software that you have never used before. What kind of user training should your company provide to you before you...
-
You have just started your first job as a financial analyst for a large stock brokerage company. Your boss, a senior analyst, has finished a detailed report evaluating bonds issued by two different...
-
Look again at Targets 2010 financial statements contained in its 2010 annual report. For instructions on how to access the report online, see the Continuing Financial Statement Analysis Problem in...
-
Find the volumes of the solids in Problems 4952 correct to the nearest unit. 4 cm -6 cm 8 cm
-
As part of a study on student loan debt, a national agency that underwrites student loans is examining the differences in student loan debt for undergraduate students. One question the agency would...
-
Show the list of all file names from a folder.
-
Which of the following is not a control activity? a. Mandatory vacations b. Risk assessment C. Security measures d. Proper authorization
-
A stock has a beta of 1.35 and an expected return of 16%. A risk-free asset currently earns 4.8%. 1. What is the expected return on a portfolio that is equally invested in the two assets? 2. If a...
-
12. A Eurodollar loan is a(n): a. ECU-denominated loan issued by a U.S. bank. b. dollar-denominated loan payable to a European bank. c. ECU-denominated loan that is subject to banking regulations in...
-
Wesley gave his 16-year old daughter, Vivian, several shares of Telus, a taxable Canadian corporation. During the year, Vivian received eligible dividends of $300 from Telus. She sold some of the...
-
(20%) A Fabry-Perot resonant cavity consists of a thin glass plate that has a refractive index of n = 1.50 and a thickness of = 100 m. Its surfaces are coated such that its peak transmittance is 100%...
-
Suppose the domestic price level increases by 15%, the exchange rate of domestic currency per foreign currency increases by 27%, and the foreign price level increases by 3%. What should be the...
-
s tart thinking about the current world of HRIS and how we've all had to adapt recently. just thinking and talking about current events and relating them back to HRIS and HR/IT/Management decisions....
-
Five standard iron solutions were prepared, and the absorbance of the solutions were measured using a spectrophotometer. The table below gives concentrations of iron ( [ Fe ] in g M L ) and their...
-
Reflecting on the book Who Gets Promoted, Who Doesn't, and Why (Asher, 2008), answer the following questions in depth. No matter what you have done in the past, the boss really doesn't care. Why?...
-
Webmasters.com has developed a powerful new server that would be used for corporations Internet activities. It would cost $10 million at Year 0 to buy the equipment necessary to manufacture the...
-
A loading condition on a beam is shown in the figure. Below are the values to be used for the various loads, concentrated forces and distances. % = 10 N/m F = 100 N a = 1 m Create a shear force and...
-
Find the reduced echelon form of each of the matrices given in Problems 120. c 1 26 + 4
-
Service-oriented architecture (SOA) is a recent approach to systems development and implementation that has much in common (and some differences, as well) with enterprise architecture. Go online and...
-
Should a Web browser be integrated into an operating system? Why or why not?
-
1. What are some of the benefits afforded to organizations implementing voice recognition technologies in these settings? How can you quantify these benefits to assess the value of the investment?...
-
You have been approached by promoters to give an opinion on the financial statements to be included in the prospectus of a proposed corporation to be named U-Park Corporation. U-Park will own and...
-
The following questions relate to the auditor's responsibility for reporting on inconsistency of application of accounting principles. Select the best response. a. Raider uses the last-in, first-out...
-
J.O. Cole, a sole proprietor, operates a trucking business. Cole also has assets and liabilities in connection with other activities. You are retained to audit the accounts of the J.O.C. Truck Lines...

Study smarter with the SolutionInn App


