You work at a chemical plant that is redesigning its unit operations to save space. One operation
Question:
You work at a chemical plant that is redesigning its unit operations to save space. One operation where liquid Neopentane at 25 degrees Celsius,1 atm, 1.5 mol/hr is mixed with liquid Carbon Disulfide at 0.5 mol/hr also 25 degrees Celsius, 1 atm is looking to be moved next to a highly sensitive chemical reactor tank. The plant manager is concerned that the enthalpy of mixing will disrupt the tank in direct contact with the outflow of the mixing operation. Another engineer, who is an expert in heat transfer, is not concerned, stating that the reaction, which operates at 25 degrees Celsius, can tolerate a change in temperature of ± 2 degrees Celsius from the outflow and not be affected.
- Assuming the operation is at a steady state and the inlet streams of Neopentane and Carbon Disulfide are well mixed, what are the activity coefficients for Neopentane and Carbon Disulfide? Use Regular Solutions Theory and Table 9.6-1 in the book (shown below) and assume the engineer is correct and Tout is approximately 25 degrees Celsius. Remember to keep your units straight!
- Calculate the rate of enthalpy generation from mixing using information from part A. (Hint: units should be in Joules/hr)
- Using the answer from Part B, calculate the temperature change of the outlet pipe and determine if there is anything to worry about. For simplicity, assume heat capacity does not change with temperature and the heat capacity of the mixture is a weighted average of the individual components.
Cp (Neopentane) = 120 J/mol-K
Cp (Carbon Disulfide) = 79 J/mol-K
Also note that ΔT = -
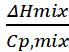 in other words, positive enthalpy means endothermic and a decrease in temperature!)
in other words, positive enthalpy means endothermic and a decrease in temperature!)
If Regular Solutions Theory was not valid and there was an increase in excess entropy of mixing, what would happen to our estimate of temperature change? Would it increase or decrease or stay the same?
Can you estimate activity coefficients and temperature change without assuming the temperature is constant? Be sure to list what assumptions you need to make to solve this.

Financial Reporting Financial Statement Analysis and Valuation a strategic perspective
ISBN: 978-1337614689
9th edition
Authors: James M. Wahlen, Stephen P. Baginski, Mark Bradshaw




