As shown in Fig. 16-2, two flasks are connected by an initially closed stopcock. One flask contains
Question:
As shown in Fig. 16-2, two flasks are connected by an initially closed stopcock. One flask contains krypton gas at 500 mmHg, while the other contains helium at 950 mmHg. The stopcock is now opened so that the gases mix. What is the final pressure in the system? Assume constant temperature.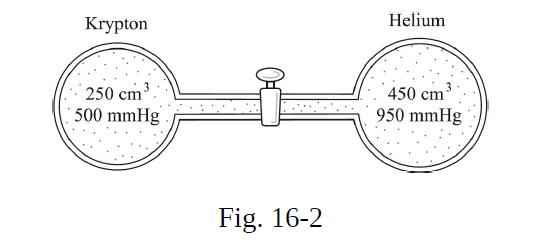
Transcribed Image Text:
Krypton 250 cm 500 mmHg 여 Fig. 16-2 Helium 450 cm 950 mmHg
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
According to Boyles Law the pressure of a gas is inversely proportional to its volume at constant ...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Physics questions
-
In what ways is this issue similar to and different from controversies about using illegal aliens to perform low-wage jobs inside the U.S.?
-
As shown in fig 1.7, a manometer is attached to a tank of gas in which the pressure is 104.0 kPa. The manometer liquid is mercury, with a density of 13.59 g/cm3. If g = 9.81 m/s2 and the atmospheric...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Find a vector parametrization of the intersection of the surfaces x 2 + y 4 + 2z 3 = 6 and x = y 2 in R 3 .
-
Discuss why many information technology professionals may overlook project cost management and how this might affect completing projects within budget.
-
Sunlight passing through a pinhole in a piece of paper casts an image of the Sun, as shown. The image size depends on the distance from the pinhole to the floor. If the paper with the pinhole is held...
-
Shades of Sand Spa Resort has 100 residential villas to accommodate guests. The accountant for the resort has asked you, the marketing manager, to prepare a budget for expected relaxation treatment...
-
(Term Modification with GainDebtors Entries) Use the same information as in E14-21 above except that American Bank reduced the principal to $1,900,000 rather than $2,400,000. On January 1, 2014,...
-
Consider the following undirected binary tree T with 17 vertices. a C M t K P Starting with the root vertex a, we can use Breadth-First Search (BFS) or Depth-First Search (DFS) to pass through all of...
-
A 500-mL sealed flask contains nitrogen at a pressure of 76.00 cmHg. A tiny glass tube lies at the bottom of the flask. Its volume is 0.50 mL and it contains hydrogen gas at a pressure of 4.5 atm....
-
One method for estimating the temperature at the center of the Sun is based on the Ideal Gas Law. If the center is assumed to consist of gases whose average M is 0.70 kg/kmol, and if the density and...
-
Explain how the Du Pont equation combines several ratios to obtain an overview of a business's financial condition.
-
Modify the symbol-table API to handle values with duplicate keys by having get() return an iterable for the values having a given key. Implement BST and Index as dictated by this API. Discuss the...
-
Enter the following transactions of an antiques shop in the accounts and extract a trial balance as at 31 March 2013. 2013 March 1 Started in business with 8,000 in the bank. 2 5 Bought goods on...
-
Develop a series of decision tree models to predict how likely a given university applicant will be accepted by the colleges based on the applicants sex, race, high school GPA, SAT/ACT score, and...
-
Enter the following items in the appropriate debtors accounts (i.e. your customers accounts) only; do not write up other accounts. Then balance-off each of these personal accounts at the end of the...
-
Subset the data to include only one of the three colleges. Group college applicants into clusters based on categorical variables of your choice (e.g., sex, parents education). Determine the...
-
What is the average difference between the price of name-brand soup and the price of store-brand soup? To obtain an estimate, an analyst randomly samples eight stores. Each store sells its own brand...
-
A 6-lb shell moving with a velocity ?? v0k explodes at point D into three fragments which hit the vertical wall at the points indicated. Fragments A, B, and C hit the wall 0.010 s, 0.018 s, and 0.012...
-
An object is located at a distance s 0 from a spherical mirror of radius R.Show that the resulting image will be magnified by an amount R MT = 2s, + R
-
Design a little dentists mirror to be fixed at the end of a shaft for use in the mouth of some happy soul. The requirements are (1) that the image be erect as seen by the dentist and (2) that when...
-
An LED 0.60 cm tall is on the central axis 30.0 cm in front of a convex spherical mirror. If the radius of curvature of the mirror is 12.0 cm determine the location of the image, describe it, and...
-
How do environmental factors, such as exposure to nature, impact physiological stress markers ? Explain
-
Discuss the effectiveness of pharmacological interventions in the management of extreme stress and their potential side effects?
-
What is the thermal shutdown temperature for the LM317 Adjustable DC Output?

Study smarter with the SolutionInn App


