A total of 6,000 lb/h of a liquid solution of 40 wt% benzene in naphthalene at 50
Question:
A total of 6,000 lb/h of a liquid solution of 40 wt% benzene in naphthalene at 50οC is cooled to 15οC. Use Figure 4.22 to obtain the weight of crystals and the flow rate and composition of mother liquor. Are the crystals benzene or naphthalene?
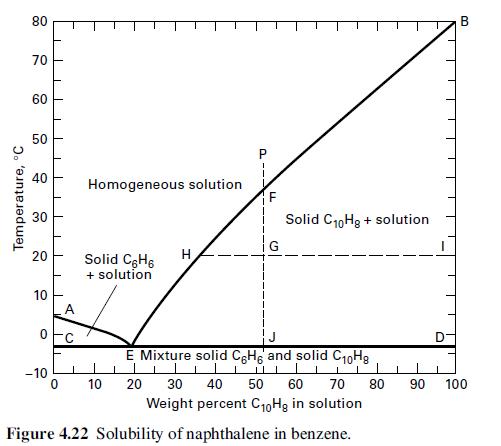
Transcribed Image Text:
Temperature, °C 80 70 60 50 40 20 10 O -10 Homogeneous solution Solid C6H6 + solution 10 H F G Solid C₁0Hg + solution B E Mixture solid CeHg and solid C10Hg ||| 1 1 20 30 40 50 60 70 80 90 100 Weight percent C10Hg in solution Figure 4.22 Solubility of naphthalene in benzene.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (10 reviews)
Solution The crystals are naphthalene The volume of t...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Separation Process Principles Chemical And Biochemical Principles
ISBN: 9780470481837
3rd Edition
Authors: By J. D. Seader, Ernest J. Henley, D. Keith Roper
Question Posted:
Students also viewed these Life Sciences questions
-
What is the probability of throwing a total of 6 points or less with three dios? n
-
A liquid containing 60 mol% toluene and 40 mol% benzene is continuously distilled in a single-equilibrium-stage unit at atmospheric pressure. What percent of benzene in the feed leaves in the vapor...
-
solution of Na2SO4. Determine the composition and flow rate of the product if the flow rate of Na2SO4 is 1275 kg/hr, and the ratio of the flow rate of the H2O to the product solution is 0.83.
-
You want to launch a business internationally, and you need to choose 3 countries1 in the Middle East, 1 in Asia, and 1 in Latin America. What are some of the components of these cultures that you...
-
Use the DemnatiRao (2004) method discussed in Exercise 23 of Chapter 9 to estimate V(tyGREG) fortyGREG defined in (11.20). Use the matrix differentiation result given in Exercise 20.
-
Calculate total average costs for the bookstore in Problem 25-3. Illustrate the store's short-run equilibrium by plotting demand, marginal revenue, average total costs, and marginal costs. What is...
-
It is said that "the higher the MARR, the higher the price that a company should be willing to pay for equipment that reduces annual operating expenses." Explain the reasoning behind this statement.
-
For seven years, Monaco Corporation has been owned entirely by Stacy and Monique, who are husband and wife. Stacy and Monique have a $165,000 basis in their jointly owned Monaco stock. The Monaco...
-
Let g be the function given by the formula g(x)=5x-3, defined for all the real (decimal) numbers x in the interval - 4x1. a. Make a table of values of g(x) for x= -4, -3, -2, -1, 0, and 1. b. Plot...
-
The comparative financial statements of Marshall Inc. are as follows. The market price of Marshall common stock was $82.60 on December 31, 20Y2. Question not attempted. Determine the following...
-
What is the secondary dew point? Is there also a secondary bubble point?
-
A mixture of chloroform (CHCl 3 ) and acetic acid at 18 C and 1 atm (101.3 kPa) is extracted with water to recover the acid. Fortyfive kg of 35 wt% CHCl3 and 65 wt% acid is treated with 22.75 kg of...
-
In 2018, Thom Inc. discovered an error in its 2015 financial statements. The firm recorded $8,500,000 of depreciation expense on its equipment instead of recording $9,500,000. Thom has a constant tax...
-
Explain the methods of transfer pricing.
-
Should group members always use the collaborating style and avoid the competing (power-forcing) style?
-
What is the six sigma approach to performance measurement.
-
Those who define others exercise control. What does this mean?
-
Explain how the creditors payment period is calculated.
-
A transformer-coupled class A amplifier drives a 16-( speaker through a 3.87:1 transformer. Using a power supply of VCC = 36 V, the circuit delivers 2 W to the load. Calculate: a. P(ac) across...
-
Three forces with magnitudes of 70pounds, 40 pounds, and 60 pounds act on an object at angles of 30, 45, and 135, respectively, with the positive x-axis. Find the direction and magnitude of the...
-
In the 1960s, the drug thalidomide was prescribed to pregnant women to treat morning sickness. However, thalidomide caused severe limb defects in the children of some women who took the drug, and its...
-
Name the compound shown below. Is this nucleotide a component of DNA, RNA, or both? Name one other function of this compound. IN HN, E 8CH 9, N. 'N. ,N 5" -0-P-0-0-P-0-0-P-0 , 2"
-
The chemical basis of blood-group specificity resides in the carbohydrates displayed on the surface of red blood cells. Carbohydrates have the potential for great structural diversity. Indeed, the...
-
Given the following algorithm for the Diffie Hellman Key Exchange and the following information as shown in Figure 1: q=23 and =7 Alice chooses X A =3 and Bob chooses Y A =6 . Calculate the secret...
-
41. According to the passage, if the Standard Presumptive Value of a vehicle is $14,655 and it is taxed the maximum allowable amount, what would be the maximum sa deduction on a tax return for the...
-
6. The conversion of nitrogen dioxide gas into dinitrogen tetroxide gas follows simple second order kinetics. a. Calculate the rate constant if the rate of the reaction is 8.49 M/sec when the...

Study smarter with the SolutionInn App


