Fifty mol% propylene in propane is separated with silica gel. The products are to be 90 mol%
Question:
Fifty mol% propylene in propane is separated with silica gel. The products are to be 90 mol% propylene and 75 mol% propane. If 1,000 lb of silica gel/lbmol of feed gas is used, can the desired separation be made in one stage? If not, what separation can be achieved? Use Figure 4.29.
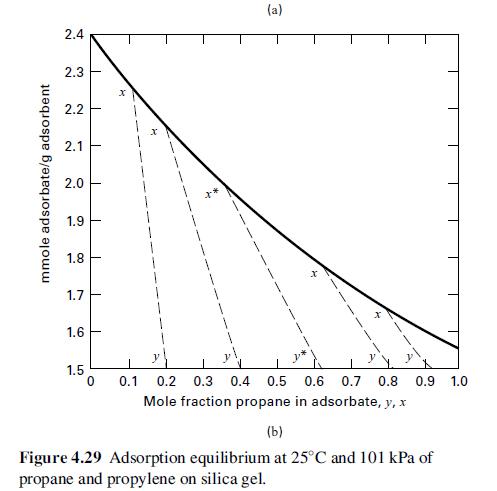
Transcribed Image Text:
mmole adsorbate/g adsorbent 2.4 2.3 2.2 2.1 2.0 1.9 1.8 1.7 1.6 1.5 T T 1 1 1 I 1 0 X (a) 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.9 1.0 Mole fraction propane in adsorbate, y, x (b) Figure 4.29 Adsorption equilibrium at 25°C and 101 kPa of propane and propylene on silica gel.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
To determine if the desired separation of 50 mol propylene in propane into 9...View the full answer

Answered By

BillClinton Muguai
I have been a tutor for the past 5 years. I have experience working with students in a variety of subject areas, including computer science, math, science, English, and history. I have also worked with students of all ages, from elementary school to college. In addition to my tutoring experience, I have a degree in education from a top university. This has given me a strong foundation in child development and learning theories, which I use to inform my tutoring practices.
I am patient and adaptable, and I work to create a positive and supportive learning environment for my students. I believe that all students have the ability to succeed, and it is my job to help them find and develop their strengths. I am confident in my ability to tutor students and help them achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Separation Process Principles Chemical And Biochemical Principles
ISBN: 9780470481837
3rd Edition
Authors: By J. D. Seader, Ernest J. Henley, D. Keith Roper
Question Posted:
Students also viewed these Life Sciences questions
-
A gas containing 50 mol% propylene in propane is to be separated with silica gel having the equilibrium properties shown in Figure. The final products are to be 90 mol% propylene and 75 mol% propane....
-
One lbmol of methane (CH4) undergoes complete combustion with stoichiometric amount of air in a rigid container. Initially, the air and methane are at 14.4 psia and 77oF. The products of combustion...
-
Propane gas is burned steadily at 1 atm pressure with a 10 percent excess oxygen supplied by atmospheric air. The reactants enter a steady flow combustor at 25C. Determine the final temperature of...
-
On January 1, 2012, Albert invested $1,000 at 6 percent interest per year for three years. The CPI (times 100) on January 1, 2012, stood at 100. On January 1, 2013, the CPI was 105; on January 1,...
-
Covariance of estimated population totals in a cluster sample. Suppose a one-stage cluster sample is taken from a population of N psus, with inclusion probabilities Ïi. Let tx and ty be the...
-
(a) Is energy dissipated in the resistor in the circuit of Figure 32.6? (b) If so, why doesn't the amplitude of the oscillations of \(v_{R}\) and \(i\) (shown in Figure 32.7) decrease with time?...
-
Toughness of a material means (a) High strength (b) High hardness (c) Ability to absorb strain energy (d) Limit of proportionality
-
Claudia Pederzolli is enrolled in a co- op program in accounting that provides an opportunity for alternating study and work terms. For her second work term, Claudia was offered a position at the...
-
Entry for Issuing Materials Materials issued are as follows: Requisition No. Material Job No. Amount 201 Aluminum 500 86,040 202 Plastic 503 29,340 203 Rubber 504 3,530 204 Glue Indirect 2,220 205...
-
Pay Corporation acquired a 75 percent interest in Sue Corporation for $600,000 on January 1, 2011, when Sue's equity consisted of $300,000 capital stock and $100,000 retained earnings. The fair...
-
A liquid of 30 mol% toluene, 40 mol% ethylbenzene, and 30 mol % water is subjected to a continuous flash distillation at 0.5 atm. Assuming that mixtures of ethylbenzene and toluene obey Raoults law...
-
A vapor mixture of equal volumes NH 3 and N 2 is contacted at 20 C and 1 atm (760 torr) with water to absorb some of the NH 3 . If 14 m 3 of this mixture is contacted with 10 m 3 of water, calculate...
-
Write a method called shift that rearranges the elements of a list of integers by moving to the end of the list all values that are in oddnumbered positions and otherwise preserving list order. For...
-
MLS fans took to social media to post pictures or videos of themselves displaying Iron Front signage in defiance of the Fan Code of Conduct. What role do you think social media played in the decision...
-
Coach Landon Donovan revealed that players intended to display an antibigotry banner during the Phoenix Rising match. If the homophobic slur had not preempted this, what effect would it have had...
-
Are White male athletes characterized differently from Black athletes and/or other athletes of color? Why or why not?
-
What kinds of examples of cause-washing do you see with MLS NEXT? What about in other brands or organizations that you support?
-
Ask someone to follow a set of instructions or to fill out a form. As an alternative, you also might test a document youve created for a course. You also may try ordering food from a website, such as...
-
The graph of y = 2(x - 3)(x - 5)(x + 4)2 has x-intercepts 3, 5, and - 4 because they are the only possible x-values that make y = 0. This is a 4th-degree polynomial, but it has only three...
-
Using a graphing utility, graph y = cot -1 x.
-
How do the RNA molecules of these two viruses differ in number and function? In your response, consider the role of both in the formation of mRNA.
-
A single virus particle of influenza A is insufficient to initiate disease in humans. It is estimated that between 100 and 1,000 influenza A virus particles are necessary to cause the flu in an...
-
Compare and contrast the reproductive life cycle of WNV (See the following figure) to that of HIV. The virus binds to a protein on the surface of a cell (not shown here) and enters the cell. e The...
-
There are 42 locations throughout the northeast region with deposits totaling $3.2bil. If you were Kiamarie's vice president, how would you expect her to choose between Gagne's Nine Events of...
-
Why is succession planning essential in an organization? 2. What is the need for succession planning? 3. What do you think are the challenges to succession planning? 4. How can employees contribute...
-
Two activities/obligations/responsibilities that might be added to an updated version of the job description for an HR professional: How would you go about monitoring changes in laws and regulatory...

Study smarter with the SolutionInn App


