For each of the following phosphors, identify the type of electronic transition responsible for luminescence and predict
Question:
For each of the following phosphors, identify the type of electronic transition responsible for luminescence and predict whether it will show weak, moderate, or strong electron–phonon coupling:
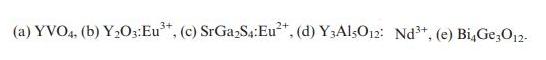
Transcribed Image Text:
(a) YVO4. (b) YO3:Eu*, (c) SrGaS4:Eu, (d) Y3AlsO12: Nd+, (e) BiG3012.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
a VO 4 3 chargetransfer transition with strong electronphonon ...View the full answer

Answered By

Nimlord Kingori
2023 is my 7th year in academic writing, I have grown to be that tutor who will help raise your grade and better your GPA. At a fraction of the cost on other sites, I will work on your assignment by taking it as mine. I give it all the attention it deserves and ensures you get the grade that I promise. I am well versed in business-related subjects, information technology, Nursing, history, poetry, and statistics. Some software's that I have access to are SPSS and NVIVO. I kindly encourage you to try me; I may be all that you have been seeking, thank you.
4.90+
360+ Reviews
1070+ Question Solved
Related Book For 

Solid State Materials Chemistry
ISBN: 9780521873253
1st Edition
Authors: Patrick M. Woodward, Pavel Karen, John S. O. Evans, Thomas Vogt
Question Posted:
Students also viewed these Sciences questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Identify whether each of the following reagents would be a strong nucleophile or a weak nucleophile, and also indicate whether it would be a strong base or a weak base: a.
-
Elastic bands are attached to tuning fork 1 ( which was 2 5 6 Hz ) to reduce its frequency. It is sounded again with tuning fork 2 ( 2 5 5 Hz ) , making 1 2 beats in 6 . 0 s . What is the new...
-
The belt sander shown in Fig.P2.35 has a belt speed of 1500 ft/min. the coefficient of friction between the sander and a plywood surface being finished is 0.2. If the downward (normal) force on the...
-
Shandra is a U.S. citizen and is the 68-year-old widowed mother of Janet. After retirement, Shandra decided to fulfill a lifelong dream and move to Paris. Shandra receives $1,000 of interest income,...
-
Consider the IRR and ERR measures of worth. If we define a root to mean a value for the measure that results in \(\mathrm{PW}=0\), then which of the following statements is true? a. Both IRR and ERR...
-
Landers Company manufactures a number of products. The standards relating to one of these products are shown below, along with actual cost data for May. The production superintendent was pleased when...
-
With what group of others do you most closely identify? Those from your home town? Those of your same sex? Religion? Race? Income level? IQ level? Who are the people with whom you feel most relaxed?...
-
As the difference Q between the equilibrium bond distances of the ground and excited states increases, would you expect the following phosphor characteristics to increase, decrease, or be unaffected:...
-
The Pr 3+ ion has 91 microstates that can be grouped into six terms before spinorbit coupling is included: 1 D, 3 F, 1 G, 3 H, 3 P, 1 S. (a) What is the electron configuration of Pr 3+ ? (b) What are...
-
a. American Express (AXP) (20%), Bank of America (BAC) (30%), JP Morgan Chase (JPM) (50%) b. AXP (20%), BAC (60%), JPM (20%) c. AXP (50%), BAC (30%), JPM (20%) d. Which portfolio would an investor...
-
How can managers cultivate a positive work culture within their team effectively?
-
Using the double-entry accounting system, show how the following transactions will generate credits and debits in the Australian Balance of Payments (BoP) accounts. Please specify clearly which...
-
Find the following values. Compounding/discounting occurs annually. Do not round intermediate calculations. Round your answers to the nearest cent. a. An initial $400 compounded for 10 years at 3%. $...
-
On April 1, 2022, Lester Company received a bank statement that showed a balance of $8,950. Lester showed an $8,000 checking account balance. The bank did not return check Number 115 for $750 or...
-
How Convertible securities are desirable as financing vehicles because they can be , whereas the the only way to induce the exercise of warrants is to have a provision
-
Frederic Chopin Corporation is preparing its December 31, 2008, balance sheet. The following items may be reported as either a current or long-term liability. 1. On December 15, 2008, Chopin declared...
-
Using thermodynamic data from Appendix 4, calculate G at 258C for the process: 2SO 2 (g) + O 2 (g) 88n 2SO 3 (g) where all gases are at 1.00 atm pressure. Also calculate DG8 at 258C for this same...
-
A 0.1429 g sample of sucrose, C 12 H 22 O 11 , is burned in a bomb calorimeter. In order to produce the same temperature rise in the calorimeter as the reaction, 2353 J must be expended. a. Calculate...
-
Identify whether you would use dilute sulfuric acid or concentrated sulfuric acid to achieve each of the following transformations. In each case, explain your choice. a. b. [H,SO,] - . [H,SO,] + H20
-
Under anaerobic conditions, glucose is broken down in muscle tissue to form lactic acid according to the reaction: C 6 H 12 O 6 (s) 2CH 3 CHOHCOOH(s). Thermodynamic data at T = 298 K for glucose and...
-
Information for two alternative projects involving machinery investments follows. Project 1 requires an initial investment of $135,000. Project 2 requires an initial investment of $98,000. Assume the...
-
Your task is to write program that takes a hexadecimal bit-pattern and prints the decimal fractional value of the number. Input-Output format: Your program will take one file name as its command-line...
-
What is the output of the following Python statements? word = "Hello World" word=word.replace("Hello", "Salam Aleykoum") word = word.swapcase() print(word.count("O"))

Study smarter with the SolutionInn App


