Use the MO diagram of oxygen to determine the oxygenoxygen bond order in the peroxide ion, O
Question:
Use the MO diagram of oxygen to determine the oxygen–oxygen bond order in the peroxide ion, O22 . Will the O–O distance in peroxide be longer or shorter than in O2?
MO diagram
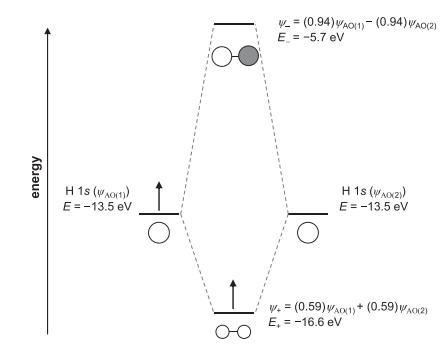
Transcribed Image Text:
energy H 1s ((1)) E = -13.5 eV Ho _= (0.94) WAO(1)- (0.94) WAO(2) E = -5.7 eV To H 1S (WA(Z)) E = -13.5 eV W+= (0.59) WAO(1) + (0.59) A0(2) E, = -16.6 eV
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Peroxide has two more electrons than O 2 Ther...View the full answer

Answered By

Ashish Bhalla
I have 12 years work experience as Professor for Accounting, Finance and Business related subjects also working as Online Tutor from last 8 years with highly decentralized organizations. I had obtained a B.Com, M.Com, MBA (Finance & Marketing). My research interest areas are Banking Problem & Investment Management. I am highly articulate and effective communicator with excellent team-building and interpersonal skills; work well with individuals at all levels.
4.80+
17+ Reviews
46+ Question Solved
Related Book For 

Solid State Materials Chemistry
ISBN: 9780521873253
1st Edition
Authors: Patrick M. Woodward, Pavel Karen, John S. O. Evans, Thomas Vogt
Question Posted:
Students also viewed these Sciences questions
-
The oxygenoxygen bond in O2+ is 112 pm and in O2 is 121 pm. Explain why the bond length in O2+ is shorter than in O2. Would you expect the bond length in O2 to be longer or shorter than that in O2?...
-
Use the molecular orbital diagram in Fig. 17.5 to determine the bond order of the Br 2 + ion. Will the BrBr bond be longer or shorter than that in the Br 2 molecule? Figure 17.5. Energy 3s 20 1 1...
-
(a) Construct a Lewis structure for hydrogen peroxide, H2O2, in which each atom achieves an octet of electrons. (b) Do you expect the O-O bond in H2O2 to be longer or shorter than the bond in O2?
-
The input file for this assignment is Weekly_Gas_Average.txt. The file contains the average gas price for each week of the year. Write a program that reads the gas prices from the file into an...
-
Determine by direct integration the product of inertia of the given area with respect to the x and y axes.
-
Find the area of the largest house Question The current selected programming language is Java. We emphasize the submission of a fully working code over partially correct but efficient code. Once...
-
Choose a country from three of the regions presented in Table 6.7. Using the Internet, collect as much information as you believe is needed to identify the potential for market segments based on age,...
-
The Lexington Company produces gas grills. This years expected production is 20,000 units. Currently, Lexington makes the side burners for its grills. Each grill includes two side burners. Lexingtons...
-
dentify the correctly revised sentence that adds interest and variety by beginning with an infinitive. Brooke congratulated me on securing investor financing for my startup company. Multiple choice...
-
Construct an MO diagram for trigonal-planar BH3 by analogy with the MOs for trigonal-planar NH 3 in Figure 5.26. Use this diagram to determine the degeneracy and orbital character of the HOMO and the...
-
What are the values of the principal and orbital angular-momentum quantum numbers for each of the following orbitals? How many radial nodes and nodal planes does each orbital possess? (a) 4s orbital,...
-
For each of the following arguments, inferring just two statements from the premises will produce a formal proof of its validity. Construct a formal proof for each of these arguments. In these formal...
-
You are working at a company in the United States. Suppose the business that you work for wants to expand its vegetarian pizza business internationally, and the CEO of the company assigned you to...
-
Laker Company reported the following January purchases and sales data for its only product. The Company uses a perpetual inventory system. Required: 2. Determine the cost assigned to ending inventory...
-
Five (5) Journal Entries are required 3. Journal entries are to be done on communication essentials lectures only. 4. Journal entries are to be typed and double spaced 5. Be sure to include on each...
-
Talk to a sports fan at least two decades older than youa relative or a friend, for instanceand ask that person to recall how being a sports fan was different when he or she was the age you are...
-
All major airlines are faced with the issue of highly volatile oil price as jet fuel is the second largest expense in the airline industry. In order to better manage its operating costs, for example,...
-
LaJolla Securities Inc. specializes in the underwriting of small companies. The terms of a recent offering were as follows: Number of shares .. 3 million Offering price ... $25 per share Net proceeds...
-
Dan and Diana file a joint return. Dan earned $31,000 during the year before losing his job. Diana received Social Security benefits of $5,000. a. Determine the taxable portion of the Social Security...
-
Using acetylene and methyl bromide as your only sources of carbon atoms, propose a synthesis for each of the following compounds: (a) (b) En Et Me En Et Me
-
Draw a mechanism and predict the major product for each reaction. a. b. c. d. e. f. ? 1) LAH H. 2) H20
-
Propose a molecular formula for a compound that exhibits the following peaks in its mass spectrum. a) (M) + at m/z = 72, relative height = 38.3% of base peak (M+1) + at m/z = 73, relative height =...
-
7. Find the decimal equivalent of the following binary numbers: [1 mark] a. (1010.0101011) = b. (10111.101) =
-
6. Find the binary equivalent for the following decimal numbers. Finfl the round off error, if there is any: [1 mark] a. (5.75)10= b. (2.625)10=
-
Consider three non-interacting electrons confined to a one-dimensional infinite potential well of width a, where V(x) = 0 for 0 x a and V(x) = otherwise. The single particle eigenfunctions including...

Study smarter with the SolutionInn App


