Question: Prove the formula (1.9.44) for the electron momentum in the case of a band described by the k p approximations (1.9.36) and (1.9.39). ():
Prove the formula (1.9.44) for the electron momentum in the case of a band described by the k · p approximations (1.9.36) and (1.9.39).
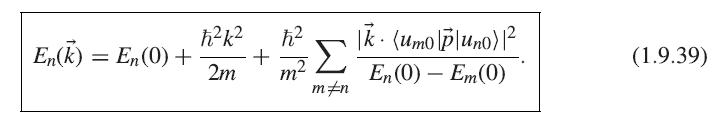
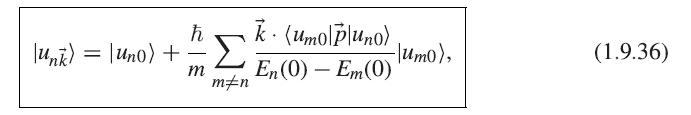
(): = m FVE, (1.9.44)
Step by Step Solution
★★★★★
3.45 Rating (168 Votes )
There are 3 Steps involved in it
1 Expert Approved Answer
Step: 1 Unlock

We write WEPWE x Unkxekz ihVeikz xUnk 1 dx unkxeik ieikikxUnk xUnk V k unk Punk Usi... View full answer

Question Has Been Solved by an Expert!
Get step-by-step solutions from verified subject matter experts
Step: 2 Unlock
Step: 3 Unlock


