Question: Using the lattice constant of iron r = 2.87 , and assuming the spins are parallel and aligned along the same axis, estimate J for
Using the lattice constant of iron r = 2.87 Å, and assuming the spins are parallel and aligned along the same axis, estimate J for the interaction (10.8.1). Then estimate the interaction J for iron using Tc = 2Jz/kB as defined in Section 10.3, assuming z = 8 for a bcc crystal, and knowing the Curie point of iron of 770° C = 1043 K. How do the two values compare?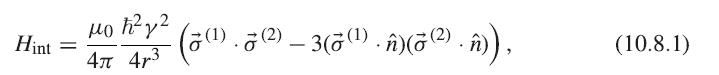
Hint 2 4 4r3 (1) (zW.(2 3W.)(2).n)), (10.8.1)
Step by Step Solution
3.40 Rating (162 Votes )
There are 3 Steps involved in it
Let 00 1 Then the vector products in the bracket in Eq 10... View full answer

Get step-by-step solutions from verified subject matter experts


