Arrange the following carbocations in order of decreasing stability. Draw all possible resonance forms for each of
Question:
Arrange the following carbocations in order of decreasing stability. Draw all possible resonance forms for each of them.
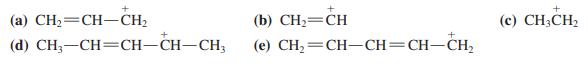
Transcribed Image Text:
(a) CH2=CH–CH2 (b) СH— СH (c) CH3CH2 (d) CH, —СH—CH-CH—CH, (e) CH,=CH-CH=CH-CH2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
No of resonance str and I effect X stability Decreas...View the full answer

Answered By

YOGENDRA NAILWAL
As I'm a Ph.D. student, so I'm more focussed on my chemistry laboratory. I have qualified two national level exams viz, GATE, and NET JRF (Rank 68). So I'm highly qualified in chemistry subject. Also, I have two years of teaching experience in this subject, which includes college teacher as well as a personal tutor. I can assure you if you hire me on this particular subject, you are never going to regret it.
Best Regards.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
List the carbocations in order of decreasing stability. a. b. CH CH;CH2CHs CH3CH2CHCH CH,CH2CH2CH2 CH,CHCH,CH, 3 CH3CHCH2CH2 CH3CHCH2CH2 Ci CH
-
Rank the following carbocations in decreasing order of stability. CH3 H CH H CH
-
List the following carbocations in decreasing order of their stability. CH +CH, +CH, CH CH
-
What is the discount yield, bond equivalent yield, and effective annual return on a $ 5 million commercial paper issue that currently sells at 98.625 percent of its face value and is 136 days from...
-
Explain why the contents of a business case might change, depending on the project.
-
The Six Sigma initiative is focused on improving communication. True or False
-
For Problem 6.8, (a) Fit the corresponding negative binomial model with the same linear predictor. (b) Compare the analysis between part (a) and that from Problem 6.8. 6.8 For the Sexual Health pilot...
-
Listed below are several types of accounting data that might be coded. For each data item, recommend a type of code (mnemonic, sequence, block, or group) and support your choice. a. Employee...
-
Supposons un consommateur ayant une richesse W qui est distribue selon une loi de densit de probabilit fw (w). Montrez qu'on peut obtenir une approximation du cot du risque, CR, en utilisant: 1 CRrr...
-
The County of Boonsville operates a jail, and some inmates work in the jail, for which they are compensated. Their earnings are deposited into an account established and held by the County in the...
-
Give the products expected from reaction of deuterium iodide (DI) with (a) 1,3-cycloheptadiene; (b) trans-1,3-pentadiene; (c) 2-methyl-1,3-pentadiene. In what way does the observable result of...
-
Sketch the molecular orbitals for the pentadienyl system in order of ascending energy (see Figures 14-2 and 14-7). Indicate how many electrons are present, and in which orbitals, for (a) the radical;...
-
Explain the difference between assurance and service warranties, and how they affect the recognition of revenue under the contract-based approach.
-
A study of fast-food nutrition compared the caloric content ofFrench Fries. Thirty-eight randomly selected servings of BurgerKing medium fries had a mean of 360 calories and a standarddeviation of 50...
-
Three Gray Ladies, Inc. had the following activities during the year. What would be reported on the Statement of Cash Flows as the net cash flow from investing activities?
-
The 20X4 Form 10-K of Dong Inc. disclosed the following: Deferred tax assets increased by $363 million and deferred tax liabilities increased by $993 million. How do these balance-sheet changes...
-
Casper Inc. issued shares of $1,000,000 in the current accounting period. How will this transaction affect the cash flow statement, if Casper uses the indirect method of calculating the cash flow?
-
Consider the following set of projects. If the firm only has $210mm in capital to invest, then which project, or projects, should be accepted? Project A PV of Cash Inflows ($mm) PV of of Cash...
-
Fred-stone Consolidated, Inc., a real estate developer, owns a 50% general partnership interest in Realty Partners, Ltd (Realty). The 50% limited partnership interests are owned by various individual...
-
The May 2014 revenue and cost information for Houston Outfitters, Inc. follow: Sales Revenue (at standard).............. $ 540,000 Cost of Goods Sold (at standard) ..........341,000 Direct Materials...
-
The chemical shift of the CH3 protons in diethyl ether is, = 1.16 and that of the CH2 protons is 3.36. What is the difference in local magnetic field between the two regions of the molecule when the...
-
Sketch the appearance of the IH-NMR spectrum of diethyl ether using J = 6.97 Hz and the data in Exercise 15.9b in a spectrometer operating at (a) 350 MHz, (b) 650 MHz.
-
Two groups of protons are made equivalent by the isomerization of a fluxional molecule. At low temperatures, where the interconversion is slow, one group has 0=5.5 and the other has 0=6.8. At what...
-
13. (8%) (a) Give a regular expression defining the same language as the following context-free grammar: S Sa, Kaa, SKb, K Ka. (b) What is the language over the alphabet (a, b) defined by the...
-
7) What is the output of the following snippet of code? myDict = { 'George': 4, 'Jean': 2, 'Paul': 10, 'Andrew': 18, 'Jacob': 8} myDict['Steve'] = 12 to Remove = ['George', 'Paul', 'Jacob'] for elt...
-
Write a program to empirically determine the performance of two power functions implementations: Iterative implementation double iterative Power (double base, int exponent) { double retVal = 1.0; if...

Study smarter with the SolutionInn App


