Question: For each synthesis in Problem 39 that does not work well, propose an alternative synthesis of the final amine, starting either with the same material
For each synthesis in Problem 39 that does not work well, propose an alternative synthesis of the final amine, starting either with the same material or with a material of similar structure and functionality.
Data From Problem 39
(a)
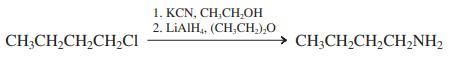
(b)
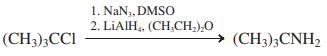
(c)
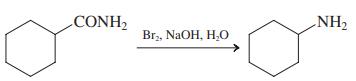
(d)
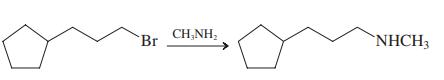
(e)
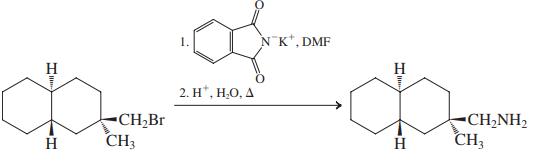
(f)
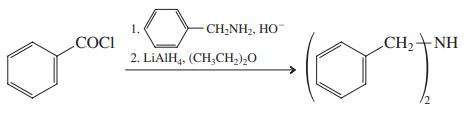
(g)
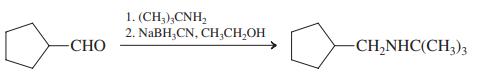
(h)
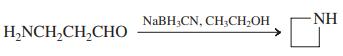
(i)
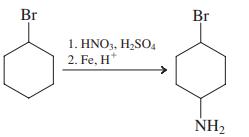
(j)
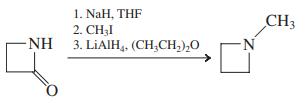
1. KCN, CH,CH, 2. LIAIH, (CH,CH,),O CH;CH2CH,CH,CI CH;CH,CH,CH,NH2
Step by Step Solution
3.35 Rating (167 Votes )
There are 3 Steps involved in it
a Alternative synthesis 1 CH3CH2MgBr then CH3OH HCl 2 NaBH4 CH3OH 3 H2SO4 heat Starting material CH3... View full answer

Get step-by-step solutions from verified subject matter experts


