Formulate a mechanism for the reaction of acetyl chloride with 1-propanol shown on p. 932. Page 932
Question:
Formulate a mechanism for the reaction of acetyl chloride with 1-propanol shown on p. 932.
Page 932
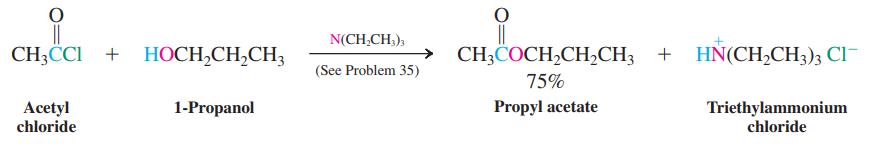
Transcribed Image Text:
N(CH,CH,), CH3CCI + HOCH,CH,CH3 → CH;COCH,CH,CH3 + HN(CH,CH3); Cl (See Problem 35) 75% Propyl acetate Acetyl chloride 1-Propanol Triethylammonium chloride
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
The reactant alcohol have two unpaired electron in oxygen atom that ...View the full answer

Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Propose a mechanism for the reaction of acetyl chloride with phenylmagnesium bromide to give 1,1-diphenylethanol. OH (1) ether solvent CH3-C CI 2 (2) H 0 acetyl chloride phenylmagnesium bromide...
-
Formulate a mechanism for the reaction described in Problem 35(b). Problem 35(b) (b) NBS (1 equivalent), hv
-
Formulate a mechanism for the reaction of butanedioic (succinic) anhydride with methanol shown on p. 935. Data From Problem 935 CH,OH H; + Alcoholysis (ester formation) 83% Propanoic anhydride...
-
Fun Cosmetics Ltd. is a skincare product manufacturer based in New Zealand that produces the LoveSkin instant moisturizer for women. Because of the reliability and quality of its products, it has...
-
To the model of U.S.-China trade presented in Section 6.2, add a third country, called Colombia. Suppose that the Colombian economy has 90 million unskilled workers and 60 million skilled workers,...
-
Why is a rollback plan necessary?
-
FRAUD PREVENTION AND DETERRENCE IN ACTION Assume the following facts: thirty-day collection period. The invoice is used to post-sales to the accounting system. Checks are received in the mailroom and...
-
Aimes Corporation's balance sheet at December 31, 2016, is presented below. During 2017, the following transactions occurred. Aimes uses a perpetual inventory system. 1. Aimes paid $2,500 interest on...
-
Researchers were curious about the size of the bill at a restaurant based on whether a credit card was used to pay versus cash. The researchers took random samples of bills at restaurants across the...
-
Lime's business has had liquidity problems for some months. The following trial balance was extracted from his books of account as at 30 September 2012: Additional information: 1. Stock at 30...
-
Give the product(s) of each of the following reactions. (a) (b) (c) (d) (e) NH2 CH;CCI + 2
-
Give the product(s) of the reactions of acetic anhydride with each of the following reagents. Assume in all cases that the reagent is present in excess. (a) (CH 3 ) 2 CHOH (b) NH 3 (c) (d) LiAlH 4 ,...
-
Some peoples everyday behavior suggests that they do not hold sunk costs irrelevant to present decisions. Give some examples different from those discussed in the chapter.
-
Assume the current interest rate on a one-year Treasury bond ( ) is 1.10 percent, the current rate on a two-year Treasury bond (R2) is 1.26 percent, and the current rate on a three-year Treasury bond...
-
Write a program called transpose.py that accepts m (int) and n (int) as command-line arguments, mxn floats from standard input representing the elements of an mxn matrix a, and writes to standard...
-
Whited Products recently completed a 4-for-1 stock split. Prior to the split, its stock sold for $120 per share. If the firm's total market value increased by 5% as a result of increased liquidity...
-
A rectangular parking lot is 5 0 . 0 ft wide and 1 4 5 ft long. What is the area of the parking lot in square meters?
-
Rabbit Company's Accounts Payable account had a balance of $22,200 on September 1, 2019, and a balance of $27,000 on September 30, 2019. During September 2019, the company made total payments of...
-
Kareem owns a pickup truck that he uses exclusively in his business. The adjusted basis is $22,000, and the fair market value is $14,000. Kareem exchanges the truck for a truck that he will use...
-
The nitrogen atoms in N2 participate in multiple bonding, whereas those in hydrazine, N2H4, do not. (a) Draw Lewis structures for both molecules. (b) What is the hybridization of the nitrogen atoms...
-
Draw all the stereo isomers of 2-bromo-3-chlorobutane and indicate whether they are enantiomers or diastereomers.
-
Label each chirality center in these compounds with an asterisk and calculate the maximum number of stereo isomers for each. CI a) b) d) c)
-
Draw all of the stereo isomers for 2, 3-dichlorobutane. Indicate which rotate plane-polarized light and which are meso.
-
You are preparing a seminar for your unit staff on effective communication. Your department supervisor has expressed interest in the seminar and wishes to review your material. You have decided you...
-
Walmart In this section you will introduce the concepts of strategic alternatives and contingency plans. Below, you will identify strategic alternatives that may increase efficiency, and contingency...
-
Inventory Turnover. Shawna s Boutique sells upscale clothing in three categories. Outerwear, Athletic wear, and Formal Wear. The annual sales and average inventory are below Annual Sales ( cost of...

Study smarter with the SolutionInn App


