From the choices shown below, pick the one that best describes the structure of compound A. Br
Question:
From the choices shown below, pick the one that best describes the structure of compound A.
Transcribed Image Text:
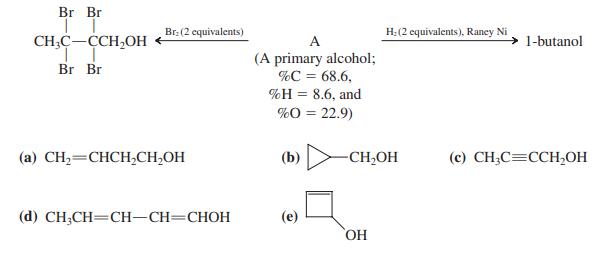
Br Br Br. (2 equivalents) H:(2 equivalents), Raney Ni CH;C-CCH,OH A 1-butanol (A primary alcohol; %C = 68.6. 9Н 3D 8.6, and %O = 22.9) Br Br (a) CH,=CHCH,CH,OH (b) -CH,OH (c) CH;C=CCH,OH (d) CH;CH=CH-CH=CHOH (e) HO,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (6 reviews)
correct ans is c 4 3 ...View the full answer

Answered By

YOGENDRA NAILWAL
As I'm a Ph.D. student, so I'm more focussed on my chemistry laboratory. I have qualified two national level exams viz, GATE, and NET JRF (Rank 68). So I'm highly qualified in chemistry subject. Also, I have two years of teaching experience in this subject, which includes college teacher as well as a personal tutor. I can assure you if you hire me on this particular subject, you are never going to regret it.
Best Regards.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
The compound whose structure is is best named (IUPAC): (a) 2-methyl-4-hexanol (b) 5-methyl-3-hexanol (c) l,4,4-trimethyl-2-butanol (d) l-isopropyl-2-hexanol (CH3),CHCH-CHCH-CH3
-
The compound whose structure is Is best named (IUPAC) (a) 3, 5-dimethylcyclopentyl ether (b) 3,5-dimethylcyclopentane-oxo (c) cis-3,5-dimethyloxacyclohexane. (d) trans-3,5- dimethyloxacyclohexane? ,...
-
The compound having the structure shown was heated with dilute sulfuric acid to give a product having the molecular formula C5H12O3 in 63-71% yield. Propose a reasonable structure for this product....
-
The following condensed balance sheet is for the partnership of Miller, Tyson, and Watson, who share profits and losses in the ratio of 6:2:2, respectively: For how much money must the other assets...
-
Describe each of the components of the PIECES framework and its uses.
-
Visit the WikiLeaks website and select two of the most current leaks posted. Evaluate the effect of the information being made public. Does it violate the privacy rights of the individuals involved?...
-
Sumitomo Cable manufactures various types of aluminum and copper cables which it sells directly to retail outlets through its distribution channels. The manufacturing process for producing cables...
-
Oxford Corporation began operations in 2012 and reported pretax financial income of $225,000 for the year. Oxfords tax depreciation exceeded its book depreciation by $40,000. Oxfords tax rate for...
-
Recent events have once again shaken and stirred the memory of the nation. Almost immediately after the Civil War, a myth arose painting the conflict with a brush that attempted to blur the lines...
-
The following data relate to the operations of Shilow Company, a wholesale distributor of consumer goods: Current assets as of March 31: Cash $ 8,000 Accounts receivable 20,000 Inventory 36,000...
-
From the choices shown below, pick the one that best describes the structure of compound A. Br Br Br. (2 equivalents) H:(2 equivalents), Raney Ni CH;C-CCH,OH A 1-butanol (A primary alcohol; %C =...
-
Draw all resonance forms and a representation of the appropriate resonance hybrid for each of the following species. CH3 () (b) () (d) ()
-
Suppose X Exp(). Find the density of Y = cX for c > 0. Describe the distribution of Y.
-
1) The proper angle for a ladder is 75 degrees from the round. Suppose you have a 15 foot ladder. How far from the household you place the base of the ladder? Round to the hundredths.(2 decimal...
-
Japan's process towards prosperity in the late 20th century was a multifaceted one, driven by a combination of domestic policies, industrial strategies, and international factors. In 1964, the...
-
The following code produces an error when executed. Why? def f1(x, y): print((x+1)/(y+1)) z=f1(3, 3)+1
-
Estimating Cost of Capital Measures Sprint Nextel Corporation (S) has $24.3 billion in total debt (which approximates its market value.) Each year this debt costs the company about $1.5 billion in...
-
Notice that the "zestimate" - Zillow's best estimate of the house price - jumps in July 2018. Can you figure out why Zillow's formula to estimate prices would predict such a large increase? Do you...
-
Roadrunner, Inc., is an exempt medical organization. Quail, Inc., a sporting goods retailer, is a wholly owned subsidiary of Roadrunner. Roadrunner inherited the Quail stock last year from a major...
-
A firm offers two products for sale. The marginal cost of one product is new zero once the first unit has been produced. The marginal cost of the other product rises as output rises. What would be...
-
The rotational constant of 12/16OC is 0.39021 cm. Calculate the bond length of the molecule (m (l2C) = 12 u exactly, m (16O) = 15.9949 u).
-
Determine the CO and CS bond lengths in OCS from the rotational constants B(16Ol2C32S) = 6081.5 MHz, B(16012C34S) = 5932.8 MHz.
-
The wave number of the incident radiation in a Raman spectrometer is 20623 cm-1. What is the wave number of the scattered Stokes radiation for the J = 4 f- 2 transition of 160O2?
-
1 . Customs officers had the right to break into the colonists' houses without their permission. This act represents a potential threat and violation of the colonists' property rights and privacy...
-
You invest $1,000 in an account that pays interest at 12 percent annual rate, compounded quarterly. How much would your investment grow to in 5 years? You plan to purchase a BMW M5 sedan for $85,000....
-
Consider Hurd Co., a U.S.-based MNC with a subsidiary in Mexico that deals in Mexican pesos (MXP). The subsidiary requires MXP200,000,000 to finance its operations over the next three years. Assume...

Study smarter with the SolutionInn App


