Give the product(s) of the reaction of butanedioic (succinic) anhydride with each of the reagents in Problem
Question:
Give the product(s) of the reaction of butanedioic (succinic) anhydride with each of the reagents in Problem 36.
Data From Problem 36
(a) (CH3)2CHOH
(b) NH3
(c)
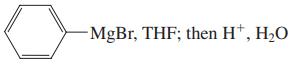
(d) LiAlH4, (CH3CH2)2O; then H+, H2O
Transcribed Image Text:
MgBr, THF; then H*, H2O
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
a b d CHCH H Succinic onhydride The opening of anhydride ...View the full answer

Answered By

Yogesh Bare
I have more than 1 yr experience in chegg, bartleby tutoring sites
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Formulate a mechanism for the reaction of butanedioic (succinic) anhydride with methanol shown on p. 935. Data From Problem 935 CH,OH H; + Alcoholysis (ester formation) 83% Propanoic anhydride...
-
Give the product(s) of reaction of cycloheptanone with each of the reagents in Problem 52. Data From Problem 52 (a) HOCH 2 CH 2 OH, H + (b) LiAlH 4 , then H + , H 2 O (c) NH 2 OH, H + (d) NH 2 NH 2 ,...
-
Give the product(s) of reaction of cycloheptanone with each of the reagents in Problem 52. Data From Problem 52 (a) HOCH 2 CH 2 OH, H + (b) LiAlH 4 , then H + , H 2 O (c) NH 2 OH, H + (d) NH 2 NH 2 ,...
-
When component Auditors are involved in the audit of group financial statements the group Auditors are required to a. consider the independence and professional reputation of the component Auditors...
-
In the model of question 44, suppose that instead of an import tariff, the government had imposed a 50% tax on exports of tea. Repeat the analysis. How do the outcomes compare with the case of the...
-
How would you use social media during the Implement phase?
-
Describe the six categories of early warning signs of potential fraud.
-
Cretin Enterprises uses a predetermined overhead rate of $21.40 per direct labor-hour. This predetermined rate was based on 8,000 estimated direct labor-hours and $171,200 of estimated total...
-
2. (7pts) Give an example of a 44 matrix A which have the following properties: (a) A is lower triangular. All its entries are integers. (b) A has two distinct eigenvalues. Each eigenvalue is a digit...
-
The following is Ash's trial balance as at 31 March 2011: Additional information: 1 Stock at 31 March 2011: 15 000. 2 At 31 March 2011 there was a specific bad debt of 6000. This was to be written...
-
Give the product(s) of the reactions of acetic anhydride with each of the following reagents. Assume in all cases that the reagent is present in excess. (a) (CH 3 ) 2 CHOH (b) NH 3 (c) (d) LiAlH 4 ,...
-
Reaction review. Suggest reagents to convert each of the following starting materials into the indicated product: (a) hexanoyl chloride into acetic hexanoic anhydride; (b) methyl hexanoate into...
-
A study published in the Journal of American Academy of Business, Cambridge (March 2002) examined whether the perception of the quality of service at five-star hotels in Jamaica differed by gender....
-
You work for a nuclear research laboratory that is contemplating leasing a diagnostic scanner. The scanner costs RM6,300,000 and it would be depreciated straight-line to zero over four years. You can...
-
Re-evaluating your Pre-Work company's IP strategy, and offering some recommendations. Have lessons learned from the course changed your views about the IP system, and how your company should interact...
-
Laurel Electronics has a quick ratio of 1.41, current liabilities of $4,462,163, and inventories of $7,863,700. What is the firms current ratio? (Round answer to 2 decimal places, e.g. 12.25.)
-
How do mitochondrial dynamics and function vary among different cell types and tissues, and how does this cellular heterogeneity contribute to organismal physiology, energy metabolism, and...
-
Calculation of individual costs and WACC Dillon Labs has asked its financial manager to measure the cost of each specific type of capital as well as the weighted average cost of capital. The weighted...
-
Louis owns three pieces of land with an adjusted basis as follows: parcel A, $75,000; parcel B, $125,000; and parcel C, $175,000. Louis sells parcel A to his uncle for $50,000, parcel B to his...
-
(a) Explain why the concentration of dissolved oxygen in freshwater is an important indicator of the quality of the water. (b) How is the solubility of oxygen in water affected by increasing...
-
Assign the configurations of the compounds represented by these Fischer projections as R or S. CH -CH2CH3 b) CH2=CH- ) >N- CH-OH CH3 Serine
-
Explain whether each of these compounds is chiral or not: a) d) H C H c=c=c CHCH CCH HC PhwSi-OCH-CH b) CHCH Ph-N-CHCH, HC Br H C CH30 CH CO H c) f) H H HC CH30 CCH CH3 COH CH3 CO H
-
Although this biphenyl is chiral, it cannot be resolved. Explain. HO-C CH30 CO H OCH
-
A company sells one model of a synthetic leather basketball. The basketballs sell for $20 each, and the variable expenses for each basketball are $14. The company has $1,200 of fixed expenses. How...
-
Below is the sales budget for Alejandro Company. The product sells for $60 in September and October and then the retail price is increased to $65 in November - February. Month Budgeted Sales in Units...
-
What is a good response to the following post made by a classmate? Interest Expense=$8,0000.09(360/36) = $72 Interest Payable (Hyatt Company) $72 (Credit) Interest Expense $72 (debit) Interest...

Study smarter with the SolutionInn App


