Name or draw the structure of each of the following compounds. (a) (b) (c) (d) (e) (f)
Question:
Name or draw the structure of each of the following compounds.
(a)
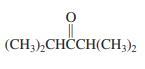
(b)
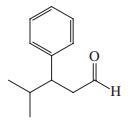
(c)
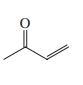
(d)
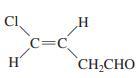
(e)
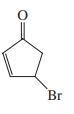
(f)
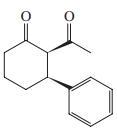
(e) (Z )-2-Acetyl-2-butenal
(f) trans-3-Chlorocyclobutanecarbaldehyde
Transcribed Image Text:
(CH3),CHCCH(CH;)2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (7 reviews)
a Name 24 Dimethylpentan 3 one b Name 4 Methyl 3 phen...View the full answer

Answered By

Binoyargha Dam
Hi everyone,
I am Binoyargha Dam, I have completed my B.Sc (Hons) and M.Sc in Chemistry and after that completed my Ph.D in organic chemistry.
I have 3 years of teaching experience (organic chemistry) in B.Sc (Hons.) course.
I also have 2 years of teaching experience in M.Tech Nanotechnology course (Polymer nanocomposites) in North-Eastern Hill University, Shillong (a central university of Govt. of India).
I have been also teaching students in coaching centers solely dedicated for preparing students for their board exams and for various entrance exams like NEET, JEE etc.
Students may feel free to contact me for any doubts related to organic chemistry, because if theisr demand matches my expertise, than I am ready to give my 100% commitment to clear their doubts in organic chemistry. I always like to share a good and friendly relation with my students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Name the following compounds: a. b. c. d. e. f. Br
-
Name the following compounds: a. b. c. d. e. f. CH3 CH3 CH2CH3 CH3 HN CH3 CH3 CH3 CH-CH3
-
Name the following compounds: (a) (b) (c) (d) (e) (f) (g) (h) CH,CH,C CH OH CH CH CH NO COOH Br OCH(CH)2 OH NO NO CH,OCH,CH,
-
Great ride (GR) is in the business of manufacturing and selling high-end vehicles. GR signs a deal with the CEO of a consulting firm for a luxury SUV. You are the long-time Controller for GreatRide...
-
Answer the following questions after reading the excerpt from Ropers v. Simmons found in Chapter 1. a. Was this a civil case or a criminal case? b. With which sources of law was the Court most...
-
Identify analyst characteristics that reduce analyst bias.
-
Increasingly, we are seeing email used in cases involving defendants located in foreign countries. Plaintiffs filed suit against four Defendants: Qingdao Sunflare New Energy Co., Skone Lighting Co.,...
-
Flowers Associates is evaluating the performance of three divisions: Daisies, Pansies, and Tulips. Using the data that follow, compute the return on investment and residual income for each division,...
-
What should you do if you are on the coast and hear a tsunami siren?
-
Values for the NASDAQ composite index during the 1,500 days preceding March 10, 2006, can be downloaded from the authors web site. Calculate the one-day 99% VaR and the one-day 99% ES on March 10,...
-
Draw structures and provide IUPAC names for each of the following compounds. (a) Methyl ethyl ketone; (b) ethyl isobutyl ketone; (c) methyl tert-butyl ketone; (d) diisopropyl ketone; (e)...
-
The following spectroscopic data are for two carbonyl compounds with the formula C 8 H 12 O. Suggest a structure for each compound. The letter m stands for the appearance of this particular part of...
-
Evaluate the integral. 5 + 1 dx (2 + 1)( 1)
-
What account(s) is/are credited when a production job is completed? Explain with example.
-
Describe a federal, state, or local policy regarding early childhood education, preferably a policy affecting your school or place of employment if your employment is a head start program.
-
How do recent pay transparency laws in several states impact corporate pay strategies? How was this surge in pay transparency influenced by Covid19 Pandemic? or was it? What other compensation trends...
-
So then given that this is a credit balance on Accounts Receivable this was already considered as your liability to the customer but why was it added back on the Trade receivables when it was the...
-
Go-Go Industries is growing at 45% per year. It is all-equity-financed and has total assets of $1 million. Its return on equity is 40%. Its plowback ratio is 55%. a. What is the internal growth rate?...
-
How can an individual taxpayer avoid having an AMT adjustment for mining exploration and development costs?
-
One study found that the elderly who do not have children dissave at about the same rate as the elderly who do have children. What might this finding imply about the reason the elderly do not dissave...
-
Show Lewis structures for the simplest neutral compounds formed from these elements: (a) Carbon and chlorine (b) Hydrogen and bromine
-
Show a Lewis structure for the simplest neutral compound formed from hydrogen and sulfur.
-
Discuss the stability of these structures: a) H H-C-O-H H b) H H-N H H
-
Give a brief description of a product launch for any business firm/organization that you want to open or would want to work with in future. Provide at least two appropriate 'Call to Actions' for the...
-
Briefly explain the organizational pattern of a 'Feature Article'? Why is 'tension' so important in a Feature article?
-
Cemex has an enviable past, a struggling present and an uncertain future. The company boasts of a century-old history and has weathered many a storm in an unfriendly Mexican business environment....

Study smarter with the SolutionInn App


