Reaction review. Without consulting the Reaction Road Maps on pp. 818 819, suggest reagents to convert
Question:
Reaction review. Without consulting the Reaction Road Maps on pp. 818 – 819, suggest reagents to convert butanal into each of the following compounds.
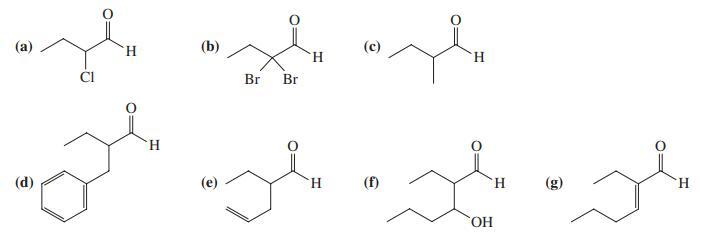
Transcribed Image Text:
(а) H. (b) (с) H. H. Br Br H. (d) H. (f) H. (g) H. HO.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
Suggest Reagents to co...View the full answer

Answered By

Samee Ullah
Algebra, Linear algebra, calculus, accounting, marketing, statistics, programming, real estate, writing, human resource management, business communication, Engineering: civil, chemical, electrical, mechanical, aerospace, building
Linguistics: sociolinguistics, applied linguistics, music, social sciences, biology, chemistry: all types, Thermodynamics, mechanics, modern physics, quantum physics, metaphysics, biology.
Feel free to contact us for all these subjects,; for quality, and best responses. Thankyou
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Reaction review II. Without consulting the Reaction Road Maps on pp. 818 819, suggest reagents to convert acetophenone into each of the following compounds. (b) C,H5 CCI3 (c) C,H5 (a) C,H5 (f) C,H5...
-
Reaction review III. Without consulting the Reaction Road Maps on pp. 818 819, suggest reagents to convert 3-buten-2-one into each of the following compounds. Page 818-819 () (b) () (d) CHs OH ()...
-
Reaction review. Without consulting the Reaction Road Map on p. 721, suggest a reagent to convert benzene into each of the following compounds. C(CH3)3 NO2 CH3 CH,CH; SO,H Br (e) (f) (h)
-
Magna Jets is a medium sized company that manufactures luxury goods for several well-known chain stores. In real terms, the company has experienced only a small growth in turnover in recent years,...
-
Connecticut made it a crime to disseminate information about birth control, even to married couples. The Supreme Court held the law unconstitutional, saying that a right to privacy was inherent in...
-
Explain why estate planning calls for collaboration between the planner and client, as well as between the client and inheritors.
-
How would the primordial helium content of the universe have been affected if the present cosmic background radiation temperature was \(27 \mathrm{~K}\) instead of \(2.7 \mathrm{~K}\) ? What about...
-
Adria Lopez, owner of Success Systems, realizes that she needs to begin accounting for bad debts expense. Assume that Success Systems has total revenues of $ 43,853 during the first three months of...
-
Q4. Show that, for all values of 8 (80), , and T> 0 there is always a positive probability that ST is negative. (Hint: consider the marginal distribution of ST.)
-
TQ 5G and Edge Computing 1- a retailer is able to track which products draw the most attention from its customers through the use of 5g-enabled motion sensors placed around the store. with this...
-
Aldol condensations may be catalyzed by acids. Suggest a role for H1 in the acid-catalyzed version.
-
A number of highly conjugated organic compounds have found use as sunscreens. One of the more widely used is 4-methylbenzylidene camphor (4-MBC), whose structure is shown in the margin. This compound...
-
Velvet Corporation has revenues of $340,000 and deductible expenses of $350,000. It also received a $40,000 dividend from a corporation in which it owns 10 percent. What is the corporations taxable...
-
Assume today is December 31, 2013. Barrington Industries expects that its 2014 after-tax operating income [EBIT(1 T)] will be $420 million and its 2014 depreciation expense will be $60 million....
-
An investor bought a bond at par and held it for one year. If the coupon rate is 5%, original maturity of the bond is 8 years, and the yield to maturity of the bond when it was sold was 6%, what is...
-
Frivolous, Inc., recently purchased an asset that will be very effective in the early years of its useful life, but the effectiveness of which will diminish significantly as maintenance charges...
-
The figure shown below is a phase diagram of the Lithium-Antimony binary system. According to the phase diagram, possible chemical reactions between Sb and Li are (1) 2Li+Sb Li2Sb or (2) 3Li + Sb ...
-
The yield on a one-year Treasury security is 5.1500%, and the two-year Treasury security has 6.950% yield. Assuming that the pure expectations theory is correct, what is the market's estimate of the...
-
Angela, who is single, incurs circulation expenditures of $153,000 during 2014. She is in the process of deciding whether to expense the $153,000 or to capitalize it and elect to deduct it over a...
-
Assume a simple Keynesian depression economy with a multiplier of 4 and an initial equilibrium income of $3,000. Saving and investment equal $400, and assume full employment income is $4,000. a. What...
-
(a) Distinguish between a step and a terrace. (b) Describe how steps and terraces can be formed by dislocations.
-
Distinguish between the following adsorption isotherms: Langmuir, BET, Temkin, and Freundlich.
-
Describe the essential features of the Langmuir-Hinshelwood, Eley-Rideal, and Mars van Krevelen mechanisms for surface-catalyzed reactions.
-
blem 3: Consider the soil profile below. If the effective stress at point C is 111 kPa, find the value of h. (30) e=0.61 Gs=2.66 Ground Surface 4m 5m e=0.48 Gs=2.67 A PP A
-
Q4 (40 marks). Two identical rigid foundations are given. Calculate the differential settlement ratio (8/L) after 100 days and long term after construction of the foundations. Hint: Average total...
-
An embankment is given (H = 5 m and y = 20 kN/m). Determine the vertical stress increase at points A, which is 5 m below the ground surface. Assume the embankment is constructed on the ground...

Study smarter with the SolutionInn App


