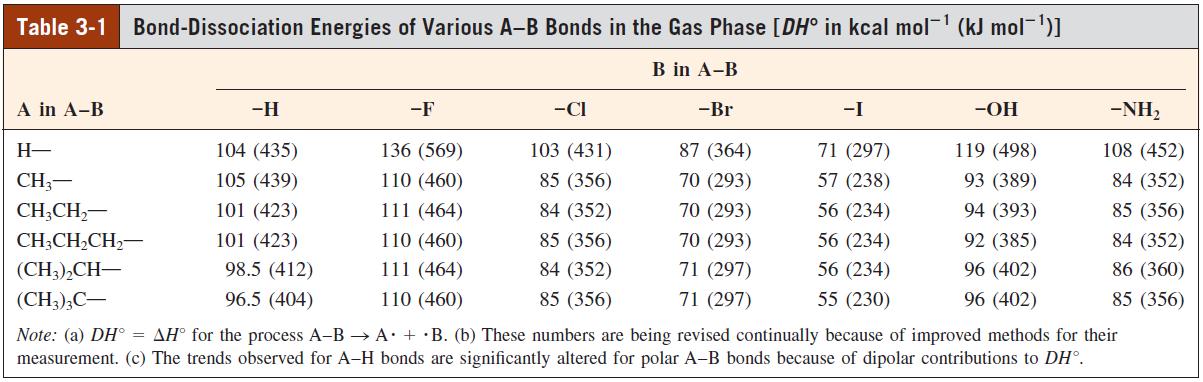
With the help of the DH values given in Tables 3-1 and 3-4, calculate the H values
Question:
With the help of the DHº values given in Tables 3-1 and 3-4, calculate the ΔHº values for addition of each of the following molecules to ethene, using 65 kcal mol-1 for the carbon – carbon π bond strength.
(a) Cl2
(b) IF (DHº = 67 kcal mol-1)
(c) IBr (DHº = 43 kcal mol-1)
(d) HF
(e) HI
(f) HO – Cl (DHº = 60 kcal mol-1)
(g) Br – CN (DHº = 83 kcal mol-1; DHº for Csp3 – CN = 124 kcal mol-1)
(h) CH3S – H (DHº = 88 kcal mol-1; DHº for Csp3 – S = 60 kcal mol-1)
Transcribed Image Text:
Table 3-1 Bond-Dissociation Energies of Various A-B Bonds in the Gas Phase [DH° in kcal mol- (kJ mol-1)1 B in A-B A in A-B -H -F -CI -Br -I -OH -NH, H- 104 (435) 136 (569) 103 (431) 87 (364) 71 (297) 119 (498) 108 (452) CH3- 105 (439) 110 (460) 85 (356) 70 (293) 57 (238) 93 (389) 84 (352) CH;CH,- 101 (423) 111 (464) 84 (352) 70 (293) 56 (234) 94 (393) 85 (356) CH;CH,CH,- 101 (423) 110 (460) 85 (356) 70 (293) 56 (234) 92 (385) 84 (352) (CH;),CH- 98.5 (412) 111 (464) 84 (352) 71 (297) 56 (234) 96 (402) 86 (360) (CH3);C- 96.5 (404) 110 (460) 85 (356) 71 (297) 55 (230) 96 (402) 85 (356) Note: (a) DH° = AH° for the process A-B → A + ·B. (b) These numbers are being revised continually because of improved methods for their measurement. (c) The trends observed for A-H bonds are significantly altered for polar A-B bonds because of dipolar contributions to DH°.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
The H values for the addition of each of the molecules to ethene can be calculated by taking into account the bond energies of the reactants and produ...View the full answer

Answered By

DHRUV RAI
As a tutor, I have a strong hands-on experience in providing individualized instruction and support to students of all ages and ability levels. I have worked with students in both one-on-one and group settings, and I am skilled in creating engaging and effective lesson plans that meet the unique needs of each student.
I am proficient in using a variety of teaching techniques and approaches, including problem-based learning, inquiry-based learning, and project-based learning. I also have experience in using technology, such as online learning platforms and educational software, to enhance the learning experience for my students.
In addition to my teaching experience, I have also completed advanced coursework in the subjects that I tutor, including mathematics, science, and language arts. This has allowed me to stay up-to-date on the latest educational trends and best practices, and to provide my students with the most current and effective teaching methods.
Overall, my hands-on experience and proficiency as a tutor have equipped me with the knowledge, skills, and expertise to help students achieve their academic goals and succeed in their studies.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Calculate Ho values for the following reactions. (a) H2 + F2 2 HF; (b) H2 + Cl2 2 HC1; (c) H2 + Br2 2 HBr; (d) H2 + I2 2 HI; (e) (CH3)3CH + F2 (CH3)3CF + HF; (f) (CH3)3CH + Cl2 (CH3)3CCl + HC1;...
-
For this assignment you just have to report the values of Timer 0 registers TCNTO, TCCROA & B for the two simple questions. Q1. To create a 20us square wave with the help of 10 MHz clock, what will...
-
The Fitness Studio, Inc., with the help of its investment bank, recently issued $43.125 million of new debt. The offer price (and face value) on the debt was $1,000 per bond and the underwriters...
-
Q3: Prove: For any sets A and B, Ax B = B A ?
-
Explain what is meant by crisis management and why many organizations continue to exist in this type of environment.
-
A firm faces an elastic demand for its product. It has come to an economist to advise it on whether to lower its price. The answer she gives is: Maybe. Why is this the right answer?
-
Abbott Industries is a well-known supplier of pharmaceuticals worldwide. Founded by Dr. Wallace Abbott, the company was incorporated in 1900 after he had been developing and making pharmaceuticals...
-
Refer to the data for Beech Corporation in Exercise 812. The company is considering making the following changes to the assumptions underlying its master budget: In Exercise 812 Beech Corporation is...
-
An object moved on the xaxis from point A to point B, turned around and moved to point C in 2 seconds. Assume direction towards the right as the positive direction. The coordinates of A, B, and C are...
-
The field work for the June 30, 2019, audit of Tracy Brewing Company was finished August 19, 2019, and the completed financial statements, accompanied by the signed audit reports, were mailed...
-
What is the IUPAC name for compound B (see margin)? (a) (E)-2-Methyl-3-pentene; (b) (E)-3-methyl-2-pentene; (c) (Z)-2-methyl-3-pentene; (d) (Z)-3-methyl-2-pentene
-
The bicyclic alkene car-3-ene, a constituent of turpentine, undergoes catalytic hydrogenation to give only one of the two possible stereoisomeric products. The product has the common name cis-carane,...
-
Describe the process for identifying potential leaders in an organization.
-
How does service ensure privacy and confidentiality to children's health needs?
-
A person stands 16cm away from of a convex mirror that has a radius of curvature of 52.5cm. What is the distance between the person and his or her image?
-
The unadjusted trail balance of Beirut Company at December 31, 2018, included the following account balances. Beiruts 2018 finance statements were issued on March 31, 2019. Accounts receivable $...
-
calculate the weighted average shares outstanding (WASO). On January 1, 2016, Canyon Corporation had 500,000 shares of common stock outstanding. On April 1, the corporation issued 100,000 new shares...
-
What information must parents provide the service about their child's long term medication?
-
On the advice of her estate planner, Grace made taxable gifts of $5 million in 2011. Grace dies in late 2013 leaving a taxable estate of $1.1 million. Grace never made any taxable gifts before 2011....
-
1. Advertising for eyeglasses _________ (increases/decreases) the price of eyeglasses because advertising promotes _________. 2. An advertisement that succeeds in getting consumers to try the product...
-
Find an expression for the fugacity coefficient of a gas that obeys the equation of state pVm = RT(1 +B/Vm + C/V-1). Use the resulting expression to estimate the fugacity of argon at 1.00 am3 and 100...
-
At 298 K the standard enthalpy of combustion of sucrose is -5797 k] mol-I and the standard Gibbs energy of the reaction is -6333 k] mol ". Estimate the additional non-expansion work that may be...
-
In 1995, the Intergovernmental Panel on Climate Change (IPCC) considered a global average temperature rise of 1.0-3SC likely by the year 2100, with 2.0C its best estimate. Because water vapour is...
-
Identify which of these forms are refundable: W - 2 / 1 0 9 8 / 1 0 9 8 - T / 1 0 9 9 - R 1 0 9 5 - Oh, so why?
-
We will learn the preparation and interpretation of a cost-volume-profit graph and a profit graph. A CVP graph highlights CVP relationships over wide ranges of activity. The anticipated profit or...
-
ABC expects to maintain the same inventories at the end as at the beginning of the year. o . The estimated fixed costs for the or are they $ 2 8 8 , 0 0 0 , and the estimated variable costs per unit...

Study smarter with the SolutionInn App


