Find the frequency of the lowest vibrational mode of a diatomic molecule in terms of the parameters
Question:
Find the frequency of the lowest vibrational mode of a diatomic molecule in terms of the parameters of the Morse potential, eq.
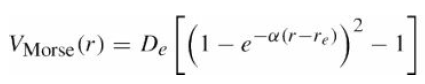
Transcribed Image Text:
Vstone () = De (1-e -r)* -1 -a(r-re
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
Expand V Morse r to second order about its minimum First fi...View the full answer

Answered By

Sumit kumar
I am an experienced online essay writer with a thorough understanding of any curriculum.and subject expert at Chegg for mathematics, CS subjects..
4.90+
5+ Reviews
13+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Physics questions
-
The overall length of a piccolo is 32.0 cm. The resonating air column vibrates as in a pipe open at both ends. (a) Find the frequency of the lowest note that a piccolo can play, assuming that the...
-
For the arrangement shown in Figure P18.64, /θ = 30.0°, the inclined plane and the small pulley are frictionless, the string supports the object of mass M at the bottom of the plane, and...
-
Magneto plasma frequency use the method of Problem 5 to find the frequency of the uniform plasma mode of a sphere placed in a constant uniform magnetic field B. Let B he along the z axis. The...
-
Write the appropriate SQL statement for the following queries. The result of the queries will be checked from your computer. What privilege should a user be given to log on to the Oracle server? Is...
-
Two blocks are sliding to the right across a horizontal surface, as the drawing shows. In Case A the mass of each block is 3.0 kg. In Case B the mass of block 1 (the block behind) is 6.0 kg, and the...
-
Make scientific discussion and conclusion for a poster about bladeless wind turbines using the following information. Bladeless wind turbines have several promising applications due to their unique...
-
What kind of responsibility (attribute or operation) are personal data? Explain.
-
Vail Resorts, Inc., owns and operates five premier year-round ski resort properties (Vail Mountain, Beaver Creek Resort, Breckenridge Mountain, and Keystone Resort, all located in the Colorado Rocky...
-
Find the units digit of 329 +1112 +15. modulus n, an
-
Tribeck Company is a family-owned business in which you own 20% of the common stock and your brothers and sisters own the remaining shares. The employment contract of Tribeck's new president, Jake...
-
The power output of air conditioners is measured intons, an ancient nomenclature dating back to the days when air was cooled by blowing it over blocks of ice.A ton of air conditioning is defined to...
-
The Morse potential parameters for oxygen are given by D e = 5.211 eV, = 2.78 1, and re =1.207 . Using the result from the previous problem, estimate the energy necessary to excite the oxygen...
-
Refer to Exercise 14.135. Do Americans spend more on health care as they age? To answer the question, economists turned to the Bureau of Labor Statistics to measure how much Americans in the...
-
Consider an Sn2 reaction of NaSH in a polar aprotic solvent with (CH3)2CHCH2CH2X. What would be the relative order of reactivity for the following X substituents? I. X= I II. X=Br III. X=Cl IV. X=F...
-
Tom sold mutual fund share he had owned 3 years so that he could use the precedes to return to college. Tom is in the 15% marginal tax bracket and his capital gains from this sale were $64,300. How...
-
A car costs $27,000 in the U.S. The same car costs 32,130 Euros. Based on these data points, please answer the below (2 decimal places). (show work along with answer) What should EUR/USD be in real...
-
Cash Flow. College students often have little income and many expenses. Does this reduce or increase the importance of completing a cash-flow statement on a monthly basis? Why?
-
The Government of Canada Zero Coupon Yield curve is shown below. What does the shape over the first three years tell you about the market's consensus expectation for future spot interest rates?...
-
Calculate the vapor pressure for a mist of spherical water droplets of radius a. 1.95 10 8 m b. 2.25 10 6 m at 298 K. The vapor pressure of water at this temperature is 25.2 Torr.
-
What is the ideal number of children to have? This question was asked on the Sullivan Statistics Survey I. Draw a dot plot of the variable Children from theSullivanStatsSurveyI data set at...
-
A uniform marble rolls without slipping down the path shown in Fig. P10.80, starting from rest. (a) Find the minimum height h required for the marble not to fall into the pit. (b) The moment of...
-
Tarzan has foolishly gotten himself into another scrape with the animals and must be rescued once again by Jane. The 60.0-kg Jane starts from rest at a height of 5.00 m in the trees and swings down...
-
A block with mass m is revolving with linear speed v 1 in a circle of radius r 1 on a frictionless horizontal surface (see Fig. E10.42). The string is slowly pulled from below until the radius of the...
-
In the Heckscher-Ohlin model, as trade occurs in a capital-abundant country, increased imports will force domestic import- competing firms to decrease prices and production. Labor and capital will...
-
Why has Gatorade been so successful in the sports drink market?
-
Mohali Foods has estimated that fixed costs per month are Rs . 1 1 4 , 8 4 0 and variable costs per unit of sales is 4 2 % . For the month the business anticipates sales of Rs . 3 0 0 , 0 0 0 . What...

Study smarter with the SolutionInn App


