Prove analytically that the entropy of a two-statesystem? is maximized when p + = p ? =
Question:
Prove analytically that the entropy of a two-statesystem?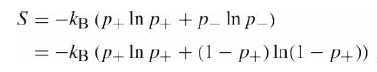
is maximized when p+ = p? = 1/2.
Transcribed Image Text:
S = -kB (p+ In p+ +p_ In p_) = -kB (P+ In p4 + (1 – P+) In(1 – P+))
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
Let p x then S k B x ln x 1 x ln1 x so at an ex...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Physics questions
-
Prove analytically that the convolution of any function (x) with a delta function, (x), generates the original function (X).
-
Show that radiation at a distribution of temperatures decreases the estimate (34.34) of the uniform temperature radiative response 0 by proving the result in the case where the radiation comes from...
-
The third law of thermodynamics states that the entropy of a perfect crystal at 0 K is zero. In Appendix 4, F-(aq), OH-(aq), and S2-(aq) all have negative standard entropy values. How can S8 values...
-
Derek and Wes take a photograph of a lake at the same angle and the same time of day. Derek's picture comes out with crisp edges around objects in the photo. Wes's picture seems to blur the outlines...
-
Before attempting this problem, review Examples 7 and 8. Two curves on a highway have the same radii. However, one is unbanked and the other is banked at an angle u. A car can safely travel along the...
-
Multinational corporations such as Gillette were widely criticized in 2017 for perpetuating the price differential between men's and women's razors. A survey by The Times in 2016 revealed that...
-
A hydraulic jack with a plunger of 30 mm diameter has a stroke of 100 mm, and a ram of 300 mm diameter. A lever arrangement with a leverage of 10 is used to drive the plunger. If a weight of 5 kN is...
-
Listed below are highway fuel consumption amounts (mi/gal) for cars categorized by the sizes of small, midsize, and large (from Data Set 14 in Appendix B). Using a 0.05 significance level, test the...
-
A glass window is 2.0 m wide and 1.5 m high and 7.5 mm thick. The temperature of the inner surface is 18 C and that of the outer surface is 11 C. (a) Find the rate of heat flow through the window....
-
A conductor with cross-sectional area of 10 cm 2 carries a conduction current 0.2 sin 10 9 tmA. Given that = 2.5 10 6 S/m and r = 6, calculate the magnitude of the displacement current density.
-
Consider an amount of helium gas at atmospheric pressure and room temperature (T = 300K) enclosed in a 1-liter partition within a cubic meter. The volume outside the partition containing the helium...
-
Consider air to be a mixture of 78% nitrogen, 21%oxygen, and 1% argon. Estimate the minimum amount of energy that it takes to separate a cubic meter of air into its constituents at STP, by computing...
-
Andrews Company uses the process cost system. The following data, taken from the organization's books, reflect the results of manufacturing operations during the month of October: Required: Prepare a...
-
Compared to using the FIFO method to account for inventory, during periods of rising prices, a company using the LIFO method is most likely to report higher: A. net income. B. cost of sales. C....
-
Intangible assets with finite useful lives mostly differ from intangible assets with infinite useful lives with respect to accounting treatment of: A. revaluation. B. impairment. C. amortization.
-
Inventory cost is least likely to include: A. production-related storage costs. B. costs incurred as a result of normal waste of materials. C. transportation costs of shipping inventory to customers.
-
Jordans response about the effect of Alphas revaluation is most likely correct with respect to the impact on its: A. return on equity. B. return on assets. C. debt to capital ratio. Brian Jordan is...
-
Carey Company adheres to U.S. GAAP, whereas Jonathan Company adheres to IFRS. It is least likely that: A. Carey has reversed an inventory write-down. B. Jonathan has reversed an inventory write-down....
-
The upper class in the 2010 survey had a household net worth between $1,345,975 and $7,402,095. Where do members of the upper class invest their money? Estimate the mean value of financial assets...
-
The following selected accounts and normal balances existed at year-end. Notice that expenses exceed revenue in this period. Make the four journal entries required to close the books: Accounts...
-
If a pilot accelerates at more than 4g, he begins to gray out but doesnt completely lose consciousness. (a) Assuming constant acceleration, what is the shortest time that a jet pilot starting from...
-
Air-Bag Injuries. During an auto accident, the vehicles air bags deploy and slow down the passengers more gently than if they had hit the windshield or steering wheel. According to safety standards,...
-
Prevention of Hip Fractures. Falls resulting in hip fractures are a major cause of injury and even death to the elderly. Typically, the hips speed at impact is about 2.0 m/s. If this can be reduced...
-
Salmon ASA has just issued a callable seven-year, 8% coupon bond with coupon payable annually. The bond can be called at par in two years or anytime thereafter on a coupon payment date. It has a...
-
Lamda corporation wants to acquire another company within its industry for $100m and it expects the acquisition to contribute to its free cash flow by $5m the first year, and this contribution is...
-
Dewan INC. has several divisions, each with a manager responsible for the operations of the division. Each division of Dewan controls product design, sales, pricing, operating costs, and profits.....

Study smarter with the SolutionInn App


