Air enters an adiabatic nozzle steadily at 300 kPa, 200C, and 45 m/s and leaves at 100
Question:
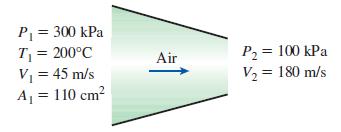
Air enters an adiabatic nozzle steadily at 300 kPa, 200°C, and 45 m/s and leaves at 100 kPa and 180 m/s. The inlet area of the nozzle is 110 cm2. Determine
(a) The mass flow rate through the nozzle
(b) The exit temperature of the air
(c) The exit area of the nozzle.
Transcribed Image Text:
P= 300 kPa T = 200°C V = 45 m/s A = 110 cm? P2 = 100 kPa V2 = 180 m/s Air %3D
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
Air is accelerated in a nozzle from 45 ms to 180 ms The mass flow rate the exit temperature and the ...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Thermodynamics An Engineering Approach
ISBN: 9781259822674
9th Edition
Authors: Yunus Cengel, Michael Boles, Mehmet Kanoglu
Question Posted:
Students also viewed these Engineering questions
-
Air enters an adiabatic nozzle steadily at 300 kPa, 200°C, and 30 m/s and leaves at 100 kPa and 180 m/s. The inlet area of the nozzle is 80 cm2. Determine (a) The mass flow rate through the...
-
Reconsider Prob. 539. Data From Q#39: Air enters an adiabatic nozzle steadily at 300 kPa, 200C, and 45 m/s and leaves at 100 kPa and 180 m/s. The inlet area of the nozzle is 110 cm 2 . Determine (a)...
-
Air enters an adiabatic nozzle steadily at 400kPa, 200oC and 35m/s and leaves at 150kPa and 180 m/s. The inlet area of the nozzle is 75cm2. Determine (a) The mass flow rate. (b) The exit temperature...
-
Allison is paid $1,520 per week. What is the amount of federal income tax withheld from Allisons paycheck under the following conditions? Use the percentage method table in the Appendix to this...
-
The following data represent the number of days absent per year in a population of six employees of a small company: 1 3 6 7 9 10 a. Assuming that you sample without replacement, select all possible...
-
What is a business model?
-
You are managing a pension fund with a goal of maximizing the long-term growth rate. There are three assets available. Asset 1 has a risk-free return of 5%. Assets 2 and 3 each are driven by...
-
The Home and Garden (HG) chain of superstores imports decorative planters from Italy. Weekly demand for planters averages 1,500 with a standard deviation of 800. Each planter costs $ 10. HG incurs a...
-
If a 15 cm diameter cast iron ball has a mass of 6.25 kg; then what diameter must a similar ball need in order to have a mass of 4.15 kg. Give your answer rounded to 3 significant digits.
-
Suppose an 18 percent drop in the price of strawberries leads to a 24 percent increase in the quantity demanded of strawberries and a 12 percent decrease in the quantity demanded of plums. a. What is...
-
Consider an adiabatic turbine operating steadily. Does the work output of the turbine have to be equal to the decrease in the energy of the steam flowing through it?
-
A house is maintained at 1 atm and 24C, and warm air inside a house is forced to leave the house at a rate of 90 m 3 /h as a result of outdoor air at 5C infiltrating into the house through the...
-
Suppose the free-fall acceleration at some location on earth was exactly \(9.8000 \mathrm{~m} / \mathrm{s}^{2}\). What would it be at the top of a \(1000-\mathrm{m}-\) tall tower at this location?...
-
As a new Tax Professional provide your financial recommendation and overview of the importance of planning financial goals and explain how a tax strategy aligns with the client's future goals. Be...
-
Fairlane Company has an ROA of 8 . 6 percent and a payout ratio of 3 3 percent. What is its internal growth rate? Do not round intermediate calculations and enter your answer as a percent rounded to...
-
Sangarwe will deposit $ 3 0 0 every quarter into an investment annuity earning 4 . 5 % compounded quarterly for seven years. What is the difference in the amount of money that she will have after...
-
How might the utilization of positive reinforcement strategies, grounded in behaviorist theory, foster a culture of continuous improvement and achievement recognition within teams and organizations?
-
A firm is trying to choose between 3 investments, all have a cost of capital of 11%. Year Project 1 Project 2 Project 3 Initial outlay $100,000 120,000 50,000 1 $ 10,000 32,000 10,000 2 20,000 32,000...
-
The financial statements of Jean Coutu are presented in Appendix B following the financial statements for Shoppers Drug Mart in Appendix A. Instructions (a) Based on the information in these...
-
A company pledges their receivables so they may Multiple Choice Charge a factoring fee. Increase sales. Recognize a sale. Collect a pledge fee. Borrow money. Failure by a promissory notes' maker to...
-
Determine the specific volume of superheated water vapor at 3.5 MPa and 4508C based on (a) The ideal-gas equation, (b) The generalized compressibility chart, and (c) The steam tables. Determine the...
-
Somebody claims that oxygen gas at 160 K and 3 MPa can be treated as an ideal gas with an error of less than 10 percent. Is this claim valid?
-
Ethane in a rigid vessel is to be heated from 50 psia and 1008F until its temperature is 540oF. What is the final pressure of the ethane as predicted by the compressibility chart?
-
Determine the exact solution of the system given below using addition or subtraction of matrices or scalar multiplication 5x - ey + 3x+2y- z = -2 . = 1 0 4x-7y+ 5z =
-
We need to be able to describe systems to assist with development, to assist with use, and to assist with maintenance. To that end, diagrams provide a concise, semi-formal way of describing a system....
-
Determine when the parametric curve x = 31261+2 and y = 4t - is concave up.

Study smarter with the SolutionInn App


