Determine the temperature at which 10 percent of diatomic hydrogen (H 2 ) dissociates into monatomic hydrogen
Question:
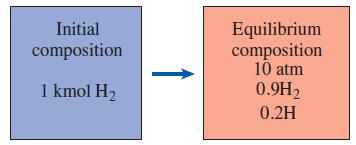
Determine the temperature at which 10 percent of diatomic hydrogen (H2) dissociates into monatomic hydrogen (H) at a pressure of 10 atm.
Transcribed Image Text:
Initial Equilibrium composition 10 atm 0.9H2 composition 1 kmol H2 0.2H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 45% (11 reviews)
The temperature at which 10 percent of H 2 dissociates into 2H is to be determined Assumptions 1 The ...View the full answer

Answered By

Kalyan M. Ranwa
I have more than seven years of teaching experience in physics and mechanical engineering.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Thermodynamics An Engineering Approach
ISBN: 9781259822674
9th Edition
Authors: Yunus Cengel, Michael Boles, Mehmet Kanoglu
Question Posted:
Students also viewed these Engineering questions
-
Determine the temperature at which 5 percent of diatomic oxygen (O2) dissociates into monatomic oxygen (O) at a pressure of 3 atm.
-
Determine the temperature at which 5 percent of diatomic oxygen (O2) dissociates into monatomic oxygen (O) at a pressure of 3 atm.
-
A mixture of 1 mol of H2 and 1 mol of Ar is heated at a constant pressure of 1 atm until 15 percent of H2 dissociates into monatomic hydrogen (H). Determine the final temperature of the mixture.
-
101, 115, 143, 106, 100, 142, 157, 163, 155, 141, 145, 153, 152, 147, 143, 115, 164, 160, 147, 150 (90%) Find the confidence interval of the median, indicated in parentheses, for the set of data.
-
The amount of milk obtained from a cow
-
Find a formula for the indicated perimeter or area. The perimeter of the figure in Fig. 2.89. A quarter-circle is attached to a triangle. Fig. 2.89. b a r
-
Consider a European Call option with strike \(K\) and time to expiration \(T\). Denote the price of the call for \(C(S, T)\) and let \(B(T)\) the price of one unit of a zero coupon bond maturing at...
-
The actual cash received from cash sales was $11,279, and the amount indicated by the cash register total was $11,256. a. What is the amount deposited in the bank for the days sales? b. What is...
-
Pharoah Industries produces and sells a cell phone-operated home security control systems. Information regarding the costs and sales during May 2022 is as follows. Unit selling price $45.00 Unit...
-
The number of items, N, produced each day by an assembly-line worker, t days after an initial training period, is modelled by N = 100 100e 0.4t (1) Calculate the number of items produced daily (a) 1...
-
The equilibrium constant for C + 1/2 O 2 CO 2 reaction at 100 kPa and 1600 K is K p . Use this information to find the equilibrium constant for the following reactions at 1600 K. (a) C + 1/2 O 2 CO...
-
At what temperature will oxygen be 15 percent disassociated at (a) 3 psia (b) 100 psia?
-
Hedged Forecasted Purchase Mansfield Corporation purchases merchandise from a German supplier on a regular basis. On April 1, 2013, Mansfield purchased 14,000 for delivery on June 30, 2013, in...
-
what ways does globalization contribute to the homogenization of cultures and the spread of Western cultural values, while also fostering hybridization, localization, and resistance to cultural...
-
Please Read The Internet and Public Policy: CriminalActivity on the Internet. Please summarize and indicate if you see anything here that will be changed by the Van Buren case ? The Internet and...
-
EMPLOYEE ID DATE OF HIRE DEPARTMENT PERFORMANCE SCORE (1-4) 485607 8/6/1987 Account Planning 1 393786 8/7/1987 Account Planning 3 355232 8/8/1987 Account Planning 3 420444 8/9/1987 Account Planning 3...
-
Question 1: Zhang Li, who runs Li's Remedies, a specialised Chinese herbs and medicines business in Anhui Province, agreed to sell 100 packets of mixed herbs and berries (Remedy Mix, 200 grams per...
-
discuss the effects of globalization on global inequality, including the concentration of wealth and power in the hands of a few, the persistence of poverty and marginalization in certain regions,...
-
What are the four steps of the professional judgment framework and how is a useful tool? What are some common judgment traps?
-
1) The government decided to reduce taxes on fast-food to increase revenue. The government assumes that fast-food products have a) An inelastic demand b) An elastic demand c) A demand curve that is...
-
A constant-volume tank contains a mixture of 120 g of methane (CH4) gas and 600 g of O2 at 25oC and 200 kPa. The contents of the tank are now ignited, and the methane gas burns completely. If the...
-
Reconsider Prob. 15-65. Using EES (or other) software, investigate the effect of the final temperature on the final pressure and the heat transfer for the combustion process. Let the final...
-
One lbmol of methane (CH4) undergoes complete combustion with stoichiometric amount of air in a rigid container. Initially, the air and methane are at 14.4 psia and 77oF. The products of combustion...
-
Explore the concept of principal quantum numbers in quantum mechanics and atomic structure. How do principal quantum numbers, denoted by the symbol "n," define the energy levels and electron...
-
1. How many electrons make up a charge of 1.0 MC? 2. What is the total charge on 1.0 kg of electrons?
-
What frequency of sound would have a wavelength the same size as a 0.81 m -wide window? (The speed of sound is 344m/s at 20 o C .)

Study smarter with the SolutionInn App


