Consider the pistoncylinder arrangement shown in the sketch below. The gas forces on each side of the
Question:
Consider the piston–cylinder arrangement shown in the sketch below. The gas forces on each side of the piston, assuming there are no frictional forces at the piston–cylinder interface, balance the weight of the piston. Determine the absolute pressure of the air (in psia) and the mass of air in the cylinder (in lbm). The atmospheric pressure is 14.6 psia.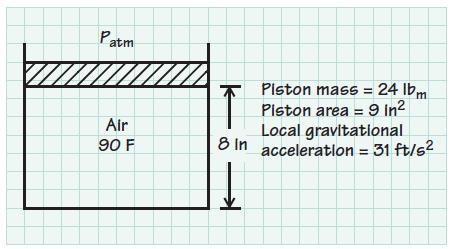
Transcribed Image Text:
Patm Air 90 F Piston mass = 24 lbm Piston area = 9 In² Local gravitational 8 In acceleration = 31 ft/s² K ➜
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 87% (8 reviews)
Given Find Mass of air in pistoncylinder and ...View the full answer

Answered By

Zablon Gicharu
I am an educator who possesses the requisite skills and knowledge due to interacting with students for an extended period. I provide solutions to various problems in step-by-step explanations, a well-thought approach and an understandable breakdown. My goal is to impart more straightforward methodologies and understanding to students for more remarkable achievements.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Thermodynamics Concepts And Applications
ISBN: 9781107179714
2nd Edition
Authors: Stephen R. Turns, Laura L. Pauley
Question Posted:
Students also viewed these Engineering questions
-
Hot air (at 50 C) is contained in a pistoncylinder arrangement as shown in the sketch for Problem 2.54. The volume of the air is 250 cm 3 . The piston has a diameter of 4 cm and a mass of 49 kg. The...
-
Hot air is contained in a pistoncylinder arrangement as shown in the sketch for Problem 2.54. The specific volume of the air is 0.25 m 3 /kg. The piston has a diameter of 6 cm and a mass of 95 kg....
-
Hot air is contained in a pistoncylinder arrangement as shown in the sketch for Problem 2.54. The density of the air is 3.6 kg/m 3 . The piston has a diameter of 5 cm and a mass of 110 kg. The gas...
-
The SEC criticized Frank Sinopoli for not sending an audit inquiry letter to Geo Securities external legal counsel. Describe the nature and purpose of such a letter. Do you agree with the SEC that...
-
National Service Company experienced the following accounting events during 2016: 1. Paid $4,000 cash for salary expense. 2. Borrowed $8,000 cash from State Bank. 3. Received $30,000 cash from the...
-
Knowledge of the law is a business asset. How can you acquire this asset short of becoming a lawyer? How is ignorance of the law a liability?
-
A New York firm is offering a new financial instrument called a "happy call." It has a payoff function at time \(T\) equal to \(\max (.5 S, S-K)\), where \(S\) is the price of a stock and \(K\) is a...
-
A popular product of Loring Glassworks is a hand-decorated vase. The companys standard cost system calls for 0.75 hours of direct labor per vase, at a standard wage rate of $8.25. During September,...
-
Individual taxt payer senario: Jhon a freelance graphic designer, earned $ 8 0 , 0 0 0 i n the last financial year. H e has expenses amounting t o $ 2 0 , 0 0 0 . Calculate his taxable income, c o s...
-
After the dot-com business he tried to start folded, David "Marty" Dawkins decided to pursue his boyhood dream of owning a bike factory. After several false starts, he finally got the small company...
-
A pistoncylinder assembly (see the above sketch) contains 0.006m 3 air at 20 C. The gas forces on each side of the piston, assuming there are no frictional forces at the pistoncylinder interface,...
-
The temperature of an ideal gas remains constant while the pressure changes from 101 kPa to 827 kPa. If the initial volume is 0.08m 3 , what is the final volume?
-
Consider the experiment of selecting two items (without replacement) from a sample space of 100 , of which 5 items are defective. Let \(A_{1}=\{\) first item selected is defective \(\}\) and...
-
You are making and designing investment portfolios for two unrelated investors.Each has $2,000,000 to invest and you can offer them shares in one or more of the four real estate investments shown in...
-
The provincial government pledged $115,000 a year for operating costs. $100,000 of the grant was advanced throughout the year. Upon receipt of the association's annual report, the government will...
-
Parkinson Plc is considering a major investment project in a new product, the Legend. Market research indicates that the demand for the Legend at a unit selling price of 12.00 and advertising costs...
-
You are given that the fair price to pay at time for a derivative paying X at time T is V = e)E[X|F], where Q is the risk-neutral probability measure and F, is the filtration with respect to the...
-
Company X has just issued some 5-year zero-coupon bonds. A continuous-time two-state model is to be used to model the status of the company and to calculate the fair price of the bonds. It is...
-
You are the auditor in charge of the audit of the municipality of Sackville, New Brunswick. The municipality has a budget of about $65 million and has had a balanced budget for the last three years....
-
What types of questions can be answered by analyzing financial statements?
-
List three products that can be used equally well by people with and without visual impairments and explain why.
-
Imagine you are tasked with designing a single dishwasher for both the European and American markets. Determine a set of global, social, environmental, and economic issues you would have to consider...
-
Find a product specification sheet for a consumer product such as an automobile, appliance, TV, motor, or something similar, and determine whether the specifications are easy to interpret. For...
-
The table below shows a recent graduate's credit card balance each month after graduation. Month 1 2 3 Debt ($) 620.00 761.88 899.80 a) Look at the graph on your calculator. Based on this graph, what...
-
Speculate sales : Speculate on the projected sales. Justify your product or service by the numbers; discuss your projected revenue gain. Note: it MUST have an ROI that justifies the project for...
-
8.6 In Figure P8.6 the experimental unit step response of a process system is shown. (a) What is the transfer function that can be used to best describe the system dynamics among the following. 1 (1)...

Study smarter with the SolutionInn App


