For the paths indicated in the sketch, show that the entropy change for an ideal gas by
Question:
For the paths indicated in the sketch, show that the entropy change for an ideal gas by either path is the same.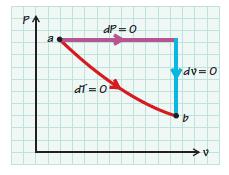
Transcribed Image Text:
PA dP=0 dl=0° dv=0 b A
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
The entropy change of an ideal gas undergoing a reversible process can be calculated using the equat...View the full answer

Answered By

Joshua Marie Geuvara
I am an academic writer with over 5 years of experience. I write term papers, essays, dissertations, reports, and any other academic paper. My main objective is to produce a high-quality paper free from plagiarism and ensure a student scores an A+. Being a fluent English speaker, I have great communication skills that also enable me to produce excellent papers.
I am conversant with most academic referencing styles (APA, MLA, and Harvard).
You can trust me with your paper and expect nothing less than quality and excellent results. I look forward to meeting with you and, more importantly, developing something that will both make us happy and satisfied.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Thermodynamics Concepts And Applications
ISBN: 9781107179714
2nd Edition
Authors: Stephen R. Turns, Laura L. Pauley
Question Posted:
Students also viewed these Engineering questions
-
Using the equation for the entropy change of an ideal gas when the volume and temperature change and TV 1 is a constant, show explicitly that the entropy change is zero for a quasi-static adiabatic...
-
Obtain the relation between the molar entropy change of an ideal gas vapor mixture and the molar entropy of each constituent.
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
If accounting-based performance measures, such as net income or core earnings, are to be significant components of manager compensation plans, they need to be sensitive and precise measures of...
-
Use the information in Exercise to prepare an October statement of retained earnings for Real Answers. In Exercise, On October 1, Keisha King organized Real Answers, a new consulting firm. On October...
-
When a measurement is very, very small or very, very large, the measurement can be expressed in scientific notation. Another option is to express the measurement using a metric prefix. For example...
-
The air velocity in the duct in Fig. P11.12 is \(750 \mathrm{ft} / \mathrm{s}\). The air static temperature is \(100^{\circ} \mathrm{F}\). Use the mercury manometer measurement to calculate the...
-
San Jose Services was formed on May 1, 2012. The following transactions took place during the first month. Transactions on May 1: 1. Jarron Gilbert invested $40,000 cash in the company, as its sole...
-
Topics and key terms have ranged from classical to contemporary and have included behavior, style, traits, job-centered,employee-centered, concern for people, concern for production, motivation,...
-
Figure 3.59 shows an exploded view of a diaphragm assembly from a gas flow meter. Determine a suitable assembly sequence for this product and carry out an analysis for manual assembly. Estimate the...
-
For a simple compressible substance with c p = a(1 + bT), where a and b are constants, determine the entropy change for an isobaric process going from T 1 and T 2 .
-
Air expands through an air turbine from inlet conditions of 690 kPa and 538 C to an exit pressure of 6.9 kPa in an isentropic process. Determine the inlet specific volume, the outlet specific volume,...
-
a. Simulate the following process for 20 days: Each day, 200 calculators are manufactured with a 5% rate of defects, and the proportion of defects is recorded for each of the 20 days. The calculators...
-
The Muamalat component of the Sharia includes: a. Commercial transactions only. b. Commercial and marriage contracts. c. Commercial, family, charitable, and hereditary laws. d. Commercial, marriage,...
-
Network performance is typically measured by which two metrics? (Choose two.) A. Bandwidth and latency B. Round Trip Time and latency C. Throughput and bandwidth D. Isolation and disaster recovery
-
How does the primary user group for government financial statements differ from that for NPO financial statements?
-
The components of Sharia include the following, except: a. Aqidah/Ibadah. b. Aqeeqah. c. Muamalat. d. Akhlaq.
-
What does the CICA Handbook consider a reasonable upper limit for the number of segments to disclose?
-
You are in the market for a new house and have decided to bid for a house at auction. You believe that the value of the house is between $125,000 and $150,000, but you are uncertain as to where in...
-
When a company has a contract involving multiple performance obligations, how must the company recognize revenue?
-
Members AB and BC can each support a maximum compressive force of 800 lb, and members AD, DC, and BD can support a maximum tensile force of 2000 lb. If a = 6 ft, determine the greatest load P the...
-
Determine the force in each member of the truss and state if the members are in tension or compression. Set P1 = 10 kN, P2 = 8 kN. F E 2 m D. A B 2 m P2
-
Determine the force in each member of the truss and state if the members are in tension or compression. Set P 1 = 8 kN, P 2 = 12 kN. F E 2 m D. A B 2 m P2
-
Three- and six-month T-Bills are trading at 0.98 and 0.96 per dollar of face, respectively. The three-month forward price of a three-month T-Bill with a face of $100 is then closest to:
-
You put $1,000 into a savings account that pays 6% in annual interest. Part 1 Attempt 1/3 for 10 pts. How much money will you have after 9 years if the account pays only simple interest? 0+ decima...
-
What is a referential integrity constraint, and why are these constraints important? What happens to our database if we don't put in our referential integrity constraints? What are other types of...

Study smarter with the SolutionInn App


