(a) Calculate the density of the atmosphere at the surface of Mars (where the pressure is 650...
Question:
(b) Compare each of these densities with that of the earth€™s atmosphere, which is 1.20 kg/m3. Consult the periodic chart in Appendix D to determine molar masses.
In Appendix D:
Periodic Table of the Elements
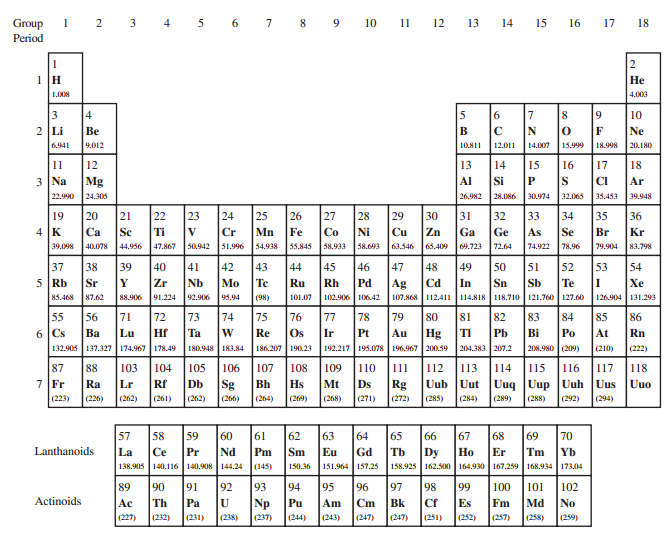
Transcribed Image Text:
4 5 6 7 8 9 10 11 12 13 14 Group 15 16 17 18 Period Не 4.003 1.008 4 2 Li 3 9. 10 Be Ne 9.012 12.011 14.007 15.999 20.180 6.941 10.811 18.998 EELE EE EEERBE 11 12 13 14 15 16 17 18 Mg 3 Na Si Al Ar 22.990 24.305 30.974 32.065 35.453 26.982 28.086 39.948 22 23 24 25 26 27 28 Fe Co 33 31 35 20 21 19 29 30 32 34 36 4 K Cr Ca Sc Mn Ni Cu Zn Ga Ge As Se Br Kr 79.904 44.956 47.867 51.996 54.938 58.933 58.693 65.409 69.723 72.64 83,798 39.098 40.078 50.942 55.845 63.546 74.922 78.96 39 47 Ag 102.906 106.42 46 50 37 38 40 41 42 43 44 45 48 49 51 52 53 54 5 Rb Nb 91.224 Mo Te Sr Zr Ru Rh Pd Cd In Sn Sb Te Xe 88.906 126.904 131.293 85.468 87.62 92.906 95.94 101.07 107.868 112.411 114.818 118.710 121.760 127.60 (98) 71 72 83 55 73 74 75 76 77 78 79 80 81 82 84 85 86 6 Cs 132.905 137.327174.967 178.49180.948 183.84 Os 192.217 195.078 196.967 200.59 204.383 207.2 Hg TI Ba Lu Hf Ta Re Ir Pt Au Pb Bi Po At Rn 208.980 (209) 186.207 190.23 (210) (222) EEE 87 7 Fr (223) 107 108 Sg (262) 88 103 104 105 106 109 110 11 112 113 14 115 116 117 118 Uub Uut Uuq Uup Uuh Ra Lr Rf Db Bh Mt Ds Rg Uus Uuo (226) (289) (294) (262) (261) (266) (264) (269) (268) (271) (272) (285) (284) (288) (292) 60 68 58 Ce 138.905 140.116140.908 144.24 (145) 61 62 65 66 Dy 158.925 162.500 164.930 167.259 168.934 173.04 69 57 59 63 64 67 70 Lanthanoids Tm La Pr Nd Pm Sm Gd Ь Er Yb 150.36 151.964157.25 90 Ac (232) 91 92 93 95 97 98 99 89 94 96 100 101 102 Actinoids Fm (257) Th Pa Np (237) Pu Am Cm Bk Cf Es Md No (247) (227) (231) (238) (244) (243) (247) (251) (252) (258) (259) EEEEEE
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (13 reviews)
Identify We know the pressure and temperature and want to find the density of the gas The ideal gas ...View the full answer

Answered By

Rinki Devi
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions.
Hi there! Are you looking for a committed, reliable, and enthusiastic tutor? Well, teaching and learning are more of a second nature to me, having been raised by parents who are both teachers. I have done plenty of studying and lots of learning on many exciting and challenging topics. All these experiences have influenced my decision to take on the teaching role in various capacities. As a tutor, I am looking forward to getting to understand your needs and helping you achieve your academic goals. I'm highly flexible and contactable. I am available to work on short notice since I only prefer to work with very small and select groups of students.
I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and helped them achieve great subject knowledge.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

University Physics with Modern Physics
ISBN: 978-0321696861
13th edition
Authors: Hugh D. Young, Roger A. Freedman, A. Lewis Ford
Question Posted:
Students also viewed these Physics questions
-
Calculate the density of oxygen at STP using the ideal gas law.
-
Calculate the density of a white dwarf whose mass is equal to the Suns and whose radius is equal to the Earths. How many times larger than Earths density is this?
-
Calculate the density of states N(E) for a metal at energy E = 8.0eV and show that your result is consistent with the curve of figure.
-
Using the Internet, explore the most useful mobile applications for a business or industry of your choice. Also explore mobile applications for this business or industry that are not currently...
-
There has been a major global crisis, and your company's board of directors has announced that the company is going bankrupt. No one could have seen this one coming. Your CEO has called you in to his...
-
The Derby Shoe Company produces its famous shoe, the Divine Loafer that sells for $ 70 per pair. Operating income for 2013 is as follows: Sales revenue ($ 70 per pair) ......$ 350,000 Variable cost...
-
The variance process in the Heston model satisfy a CIR process: \[d V_{t}=\kappa\left(\bar{V}-V_{t} ight)+\sigma \sqrt{V_{t}} d W_{t}\] Use Ito to calculate the dynamics of the volatility process...
-
A British warship fires a projectile due south near the Falkland Islands during Word War I at latitude 50oS. If the shells are fired at 37o elevation with a speed of 800 m/s, by how much do the...
-
How do emerging technologies such as biofeedback systems and virtual reality environments facilitate the implementation of personalized stress reduction interventions, and what are the potential...
-
Consider a society consisting only of Helen, who allocates her time between sewing dresses and baking bread. Each hour she devotes to sewing dresses yields 4 dresses and each hour she devotes to...
-
A cylindrical tank has a tight-fitting piston that allows the volume of the tank to be changed. The tank originally contains 0.110 m 3 of air at a pressure of 0.355 atm. The piston is slowly pulled...
-
A large cylindrical tank contains 0.750 m 3 of nitrogen gas at 27 o C and 7.50 10 3 (absolute pressure). The tank has a tight-fitting piston that allows the volume to be changed. What will be the...
-
What is the primary risk of trying to stimulate moderate levels of conflict in a situation characterized by lethargy?
-
The Bonding Company prepares accrual basis financial statements every June 30 and December 31. On 1/1/2001 the company issued bonds to the public. The bonds are due at the end of 20 years, on...
-
The Jablonski family has a monthly income of $6,500. They're applying for an FHA loan with monthly payments of $2,200. They have student loan payments of $300 per month and a minimum payment of $20...
-
In addition to knowing that the visa became current on May 1, 2023, you find out the following information: The petition was filed on August 2, 2010. It was approved on March 15, 2012. Liam was born...
-
Content AreaOmega Inc. is expecting a reduction of $25,000 in fixed costs of $725,000. What will be the change in break-even sales (in units) if selling price per unit is $50 and the unit variable...
-
Are "unconditional promises to give" required to be recognized as liabilities? 18. What is the recognition threshold for recording a gain contingency? In an internal use software project, what are...
-
Stateful packet filters maintain two data structures. Name them and briefly describe what they do.
-
How many years will it take a $700 balance to grow into $900 in an account earning 5%?
-
A student is sitting on a frictionless rotating stool with her arms outstretched as she holds equal heavy weights in each hand. If she suddenly lets go of the weights, will her angular speed...
-
If two spinning objects have the same angular momentum, do they necessarily have the same rotational kinetic energy? If they have the same rotational kinetic energy, do they necessarily have the same...
-
If the earths climate continues to warm, ice near the poles will melt, and the water will be added to the oceans. What effect will this have on the length of the day? Justify your answer.
-
Assume that a robot systems constant failure and repair rates are 0.0004 failures per hour and 0.002 repairs per hour, respectively. Calculate the robot system steady-state unavailability and...
-
March 6, 2018 David Beckham's Latest Pitch: Men's Cosmetics. By Robert Williams Men of the world, David Beckham has a message for you: Moisturize. "It's the biggest thing," he says in a London...
-
3. On January 1, an investment fund was opened with an initial balance of 5000. Just after the balance grew to 5200 on July 1, an additional 2600 was deposited. The annual effective yield rate for...

Study smarter with the SolutionInn App


