Question: As shown in Table 24.1, water has a very large dielectric constant K = 80.4. Why do you think water is not commonly used as
As shown in Table 24.1, water has a very large dielectric constant K = 80.4. Why do you think water is not commonly used as a dielectric in capacitors?
Table 24.1
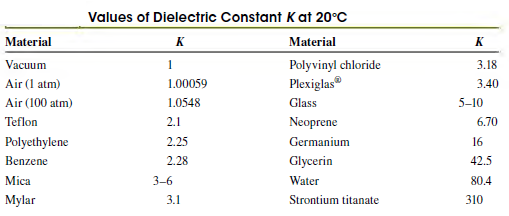
Values of Dielectric Constant Kat 20C Material Material Polyvinyl chloride Plexiglas Vacuum 1 3.18 Air (1 atm) 1.00059 3.40 Air (100 atm) 1.0548 Glass 5-10 Teflon 2.1 Neoprene 6.70 Polyethylene 2.25 Germanium 16 Benzene 2.28 Glycerin 42.5 Mica 3-6 Water 80.4 Strontium titanate Mylar 3.1 310
Step by Step Solution
★★★★★
3.48 Rating (158 Votes )
There are 3 Steps involved in it
1 Expert Approved Answer
Step: 1 Unlock

Water is an excellent solvent and ... View full answer

Question Has Been Solved by an Expert!
Get step-by-step solutions from verified subject matter experts
Step: 2 Unlock
Step: 3 Unlock


