The binding energy per nucleon for most nuclides doesnt vary much (see Fig. 43.2). Is there similar
Question:
Fig.43.2
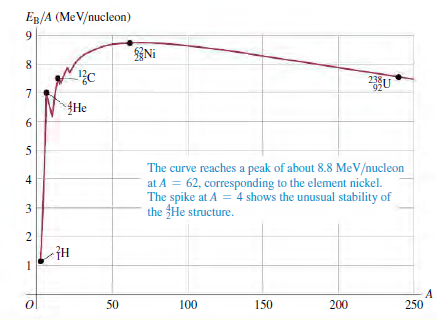
Transcribed Image Text:
ER/A (MeV/nucleon) Ni 238U 92 {He The curve reaches a peak of about 8.8 MeV/nucleon at A = 62, corresponding to the element nickel. The spike at A = 4 shows the unusual stability of the He structure. 50 100 150 200 250 6. 4) 3.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (8 reviews)
No As the number of electrons increases so does ...View the full answer

Answered By

Rinki Devi
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions.
Hi there! Are you looking for a committed, reliable, and enthusiastic tutor? Well, teaching and learning are more of a second nature to me, having been raised by parents who are both teachers. I have done plenty of studying and lots of learning on many exciting and challenging topics. All these experiences have influenced my decision to take on the teaching role in various capacities. As a tutor, I am looking forward to getting to understand your needs and helping you achieve your academic goals. I'm highly flexible and contactable. I am available to work on short notice since I only prefer to work with very small and select groups of students.
I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and helped them achieve great subject knowledge.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

University Physics with Modern Physics
ISBN: 978-0133977981
14th edition
Authors: Hugh D. Young, Roger A. Freedman
Question Posted:
Students also viewed these Physics questions
-
The binding energy per nucleon for magnesium-27 is 1.326 X 10-12 J/nucleon. Calculate the atomic mass of magnesium-27.
-
Calculate the binding energy in J/nucleon for carbon-12 (atomic mass 12.00000) and uranium-235 (atomic mass 235.0439). The atomic mass of 11H is 1.00782 amu, and the mass of a neutron is 1.00866 amu....
-
Calculate the binding energy per nucleon for a 14/7N nucleus.
-
Which of the following is not a strategic disadvantage of vertical integration? Vertical integration poses all kinds of capacity-matching problems (achieving the most efficient scale of operation for...
-
What is the rule of 78s?
-
If Mel Jones (Classroom Demonstration Exercise 2) amortized his truck by the units-of production method, calculate the first years amortization based on the following: cost $6,000; residual value...
-
Use technology to find the regression line to predict $Y$ from $X$. $X$ 2 4 6 8 10 12 $Y$ 50 58 55 61 69 68
-
Sarah Edwards, division manager for Pillows Plus, is speaking to the controller, Diana Rothman, about the budgeting process. Sarah states, Im not an accountant, so can you explain the three main...
-
Ramer and Knox began a partnership by investing $ 6 0 , 0 0 0 and $ 9 0 , 0 0 0 , respectively. The partners agreed to share net income and loss by giving annual salary allowances of $ 5 0 , 0 0 0 to...
-
Three forces are exerted on an object placed on a tilted floor in Figure P3.43. The forces are measured in newtons (N). Assuming that forces are vectors, a. What is the component of the net force...
-
Heavy, unstable nuclei usually decay by emitting an a or a b particle. Why dont they usually emit a single proton or neutron?
-
What are the six known elements for which Z is a magic number? Discuss what properties these elements have as a consequence of their special values of Z.
-
Which type of products are suitable for purchase by reverse auction?
-
A golf ball is hit from the tee and travels above level ground. Accounting for air resistance, where is the horizontal location of the peak of the flight: at a position that is less than half the...
-
A rifle is aimed horizontally at a target \(100 \mathrm{~m}\) away, and the bullet leaves the rifle barrel at \(650 \mathrm{~m} / \mathrm{s}\). If the gun is aimed right at the bull's-eye, by how...
-
You leave your house and walk east for \(1.0 \mathrm{~h}\), northeast for \(1.5 \mathrm{~h}\), south for \(1.0 \mathrm{~h}\), and southwest for \(2.5 \mathrm{~h}\), always moving at the same speed....
-
The toy race track in Figure P11.4 has three lanes, and cars cannot change lanes. The centerlines of any two adjacent lanes are 100 mm apart, and the innermost lane has a radius of 1.00 m. (a) If...
-
Your directions on a scavenger hunt map say to walk \(36 \mathrm{~m}\) east, then \(42 \mathrm{~m}\) south, then \(25 \mathrm{~m}\) northwest. With the positive \(x\) direction being east, what is...
-
A bond that settles on June 7, 2016, matures on July 1, 2036, and may be called at any time after July 1, 2026, at a price of 105. The coupon rate on the bond is 6 percent and the price is 115.00....
-
Why can wastewater treatment requirements in Hawaii be less stringent than those in most locations on the U.S. mainland?
-
Assume that a message signal is given by m(t) = 4 cos(2f m t) + cos(4f m t) Calculate an expression for x c (t) = 1/2 A c m(t) cos(2f m t) 1/2 A c m (t) sin (2f c t) for A c = 10. Show, by sketching...
-
Consider the system shown in Figure 3.35. Assume that the average value of m(t) is zero and that the maximum value of |m(t)| is M. Also assume that the square-law device is defined by y(t) = 4x (t) +...
-
An AM modulator is operating with an index of 0.8. The modulating signal is m(t) = 2 cos(2f m t) + cos(4f m t) + 2 cos(10f m t) (a) Sketch the spectrum of the modulator output showing the weights of...
-
A 1 kg metal block is heated to 200 C and then dropped into a thermally isolated container with 4 kg of water and 100 gr of ice both at a temperature of 0 C. If the specific heat of water is cw=4186...
-
Why are both ER Diagrams and Relational Modeling needed when a database system is developed?
-
Which data model provides a user with the best "program-data independence"? A. Hierarchial Model B. Network Model C. Relational Model D. Entity Relationship Model Why?

Study smarter with the SolutionInn App


