Tell how much hydrogen is bonded to each carbon in the following compounds, and give the molecular
Question:
Tell how much hydrogen is bonded to each carbon in the following compounds, and give the molecular formula of each substance:
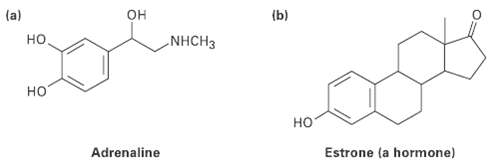
Transcribed Image Text:
(b) он NHCH3 но но но Adrenaline Estrone (a hormone)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
Strategy Remember that the end of a line represents a carbon atom with 3 hydrogen...View the full answer

Answered By

Sandip Agarwal
I have an experience of over 4 years in tutoring. I have solved more than 2100 assignments and I am comfortable with all levels of writing and referencing.
4.70+
19+ Reviews
29+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Hydrogen is bonded to fluorine in hydrogen fluoride by a covalent bond. Write a Lewis formula for hydrogen fluoride.
-
Give the molecular formula of a cyclic alkane, a cyclic alkene, alinear alkyne, and an aromatic hydrocarbon that in each case contains six carbon atoms.Which are saturated and which are unsaturated...
-
Hydrogen is bonded to fluorine in hydrogen fluoride by a covalent bond. Write a Lewis formula for hydrogen fluoride.
-
Why is it likely to have preexisting normal faults in an orogenic belt?
-
During his senior year in college, Sandy is drafted by the Los Angeles Dodgers. When he graduates, he expects to sign a five-year contract in the range of $1.7 million per year. Sandy plans to marry...
-
Discuss how you would segment students at your school. Does the student union do this?
-
Suppose at time 0 you have arranged to be paid at time \(T\) the amount \(\int_{0}^{T} S(t) \mathrm{d} t\), where \(S(t)\) is the spot price at \(t\) of a commodity that can be shorted and has zero...
-
December 31, 20X5 Posey Company acquired 90% of Stargell Corporation's outstanding common stock for $1,116,900. On that date: ? The fair value of the noncontrolling interest was $124,100; ? Stargell...
-
Two speeding lead bullets, one of mass 13.0 g moving to the right at 320 m/s and one of mass 7.95 g moving to the left at 385 m/s, collide head-on, and all the material sticks together. Both bullets...
-
You are interviewing for an entry-level financial analyst position with Wayne Industries. Bruce Wayne, the senior partner, wants to be sure all the people he hires are very familiar with basic...
-
Identify all nonbonding lone pairs of electron in the following molecules, and tell what geometry you expect for each pf the indicated atoms. (a) The oxygen atom in the dimethyl ether, CH3 ? O ? CH3...
-
Propose skeletal structures for compounds that satisfy the following molecular formulas. There is more than one possibility in each case. (a) C5H12 (b) C2H7N (c) C3H6O (d) C4H9Cl
-
Which type of radiation-alpha, beta, or gamma-predominates within an enclosed elevator descending into a uranium mine?
-
What are the top two medications that are most used to manage Type I Diabetes Mellitus?
-
Vacant Home Tax has been a point of contention across Ontario over the last couple of years. From a Municipal perspective, provide some commentary around the benefits and drawbacks of a implementing...
-
If a second-grade student is able to independently identify uppercase and lowercase receptive and color words what would be a good next thing for the child to learn?
-
From the two, laws and ethics which is more significant in influencing behavior?
-
Answer the following question on the discussion board: Some family-law attorneys post an online interview form or checklist that they ask prospective clients to fill out or be ready to discuss. Find...
-
How does the concept of federalism complicate the administration of criminal justice in the United States?
-
For the data in Exercise 17-19, use the FIFO method to summarize total costs to account for, and assign these costs to units completed and transferred out, and to units in ending work in process....
-
Compute the partial molar volumes of methyl formate in methanolmethyl formate and ethanolmethyl formate mixtures at 298.15 K for various compositions using the experimental data in Fig. 8.1-2a and...
-
Draw a structure for each model. Explain whether the molecule represented by each model has a dipole moment or not.
-
Draw a structure for each model. Show the direction of the dipole moment for the molecule represented by each model.
-
Draw a structure for the model, N the molecule polar or non-polar explain?
-
given l o g x 5 = l o g 5 x solve for x
-
Solve the formula 8 x 3 y = - 1 9 for y .
-
Q6 Find the total amount for of the current balance for each underwriter and find out top 10 richest underwriter Q7 Create a pivot table for the entire data and find out the total current balance for...

Study smarter with the SolutionInn App


