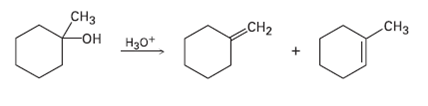
The acid-catalyzed dehydration of 1-methylcyclohexanol yields a mixture of two alkenes. How could you use 1H NMR
Question:
The acid-catalyzed dehydration of 1-methylcyclohexanol yields a mixture of two alkenes. How could you use 1H NMR to help you decide which waswhich?
Transcribed Image Text:
CHз CHз -он на0* CH2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (15 reviews)
Use of 3C NMR to distinguish between the two isomers has been described in the text in Sectio...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How could you use 1H NMR spectroscopy to distinguish among the following esters? 018 18 - CH3 OH
-
How could you use 1H and 13C NMR to help you distinguish among the following isomeric compounds of formulaC4H8? CH2-CH2 H2H CH CCH3HCH CH2-CH2 CHH2
-
How could you use 1H NMR to distinguish between the following pairs ofisomers? (a) CH3CH=CHCH2CH3 and CH2 H H2H () CH20CH2CH and CH3OCH2CH2CH3 (c) CHC,H and CH3CH2CH3 (d) HCICH)H and CH3CH=CHCH3
-
What conditions must be met for revenue to be recorded? Can pledges meet those conditions?
-
In each of the following situations why are firms likely to benefit from vertical integration? (a) A grain elevator is located at the terminus of a rail line. (b) A manufacturer of a product with a...
-
Calculate the value of ZL in the circuit of Fig. 11.48 in order for ZL to receive maximum average power. What is the maximum average power received by ZL? -j10 30 40 j20
-
For a specific paint application, a mixture of solvents is needed. The mixture will be identified by its ability to mix with water (total miscibility), its normal boiling point (determines the...
-
The comparative balance sheet of Green Earth Lawn and Garden Inc. for December 31, 2008 and 2009, is as follows: The income statement for the year ended December 31, 2009, is as follows: The...
-
The costs per equivalent unit of direct materials and conversion in the Rolling Department of Jabari Steel Company are $1.00 and $2.15, respectively. The equivalent units to be assigned costs are as...
-
McDougan Associates, a U.S.-based investment partnership, borrows 80,000,000 at a time when the exchange rate is $1.3460/. The entire principal is to be repaid in three years, and interest is 6.250%...
-
Predict the splitting pattern for each kind of hydrogen in isopropyl propanoate, CH3CH2CO2CH(CH3)2.
-
Propose structures for compounds with the following formulas that show only one peak in their 1H NMR spectra: (a) C5H12 (b) C5H10 (c) C4H8O2
-
In Problems 1-3, R = {(x, y): 0 ( x ( 6, 0 s y ( 4} and P is the partition of R into six equal squares by the lines x = 2, x = 4, and y = 2. Approximate ((x, y) dA by calculating the corresponding...
-
Many things grow exponentially. The general formula for exponential growth is: g(t) = Ae", where A is the starting value, r is the rate of growth and t is time. Assume that A = 1, and r = 1.07 (a 7%...
-
Bambazonke plans to sell 8 3 , 0 0 0 units of product number 7 9 4 in May, and each of these units requires three units of raw material. Pertinent data follow. Product Number 7 9 4 Raw Material...
-
A pipe strut BC is loaded and supported as shown. The strut has a uniform cross section and a mass of 12 kg. Determine the tension in the cable and the reaction at support C. Write your answers using...
-
Obligations imposed on state governments, by Congress, without reimbursement have come to be known as O blank checks. O assigned obligations. Ounchecked authorizations. unfunded mandates
-
Assume an issue is decided by a state supreme court. On what cases is that decision binding?
-
Solve the linear, first-order IVP. \(\frac{1}{2} \dot{x}+t x=\frac{1}{2} t, x(0)=\frac{1}{3}\)
-
A supermarket chain is interested in exploring the relationship between the sales of its store-brand canned vegetables (y), the amount spent on promotion of the vegetables in local newspapers (x1)...
-
A dioxin-contaminated water source contains 0.085% dioxin by mass. How much dioxin is present in 2.5 L of this water? Assume a density of 1.00 g/mL.
-
Show how you would synthesize butyl isopropyl sulfide using butan-1-ol, propan-2-ol, and any solvents and reagents you need.
-
Mustard gas, Cl - CH2CH2 - S - CH2CH2 - Cl,was used as a poisonous chemical agent in World War I. Mustard gas is much more toxic than a typical primary alkyl chloride. Its toxicity stems from its...
-
Show how you would use a protecting group to convert 4-bromobutan-1-ol to hept-5-yn-1-ol.
-
Excerpts from Andre Company's December 31, 2024 and 2023, financial statements are presented below: Accounts receivable Inventory Net sales Cost of goods sold Total assets Net income Total...
-
The York City Hospital has just acquired new equipment. The equipment cost $ 4 , 2 5 0 , 0 0 0 , and the organization spent $ 1 3 5 , 0 0 0 on upgrading the physical plant to the new equipment will...
-
Carmen Camry operates a consulting firm called Help Today, which began operations on December 1. On December 31, the company's records show the following selected accounts and amounts for the month...

Study smarter with the SolutionInn App


