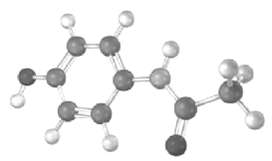
The following model is a representation of acetaminophen, a pain reliever sold in drugstores as Tylenol. Identify
Question:
The following model is a representation of acetaminophen, a pain reliever sold in drugstores as Tylenol. Identify the hydribization of each carbon atom in acetaminophen, and tell which atoms have lone pairs of electrons (gray = C, red = O, blue =H).
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
H H H CC 11 H CCsp 11 H O H Acetamino...View the full answer

Answered By

Munir Ahmed Jakhro
I am professional Tutor of of Business Courses, I did my four years Bachelor Degree from one of the Top Business schools of World "Institute of Business Administration" in year 2013. Since then I have been working as Tutor of Accounting, Finance tutor on different online platforms like this website. I am have experience of 6 years teaching business courses to students online and offline my professional job at national savings also helped me in accounting understanding .
4.90+
8+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The following model is a representation of asparatame, C14H18N2O5, known commercially as NutraSweet. Only the connection between atoms is shown; multiple bonds are not indicated. Complete the...
-
The following model is specified: y1 = 1y2 + 11x1 + 1, y2 = 2y1 + 22x2 + 32x3 + 2. All variables are measured as deviations from their means. The sample of 25 observations produces the following...
-
The following model is specified:y 1 = 1 y 2 + 11 x 1 + 1 ,y 2 = 2 y 1 + 22 x 2 + 32 x 3 + 2 . All variables are measured as deviations from their means. The sample of 25 observations produces...
-
What are the premises for successful paleostress analysis?
-
Amber Company has used the dollar-value LIFO technique for the past three years. The company has only one inventory pool. Its beginning inventory for the current year was computed as follows: a. The...
-
(+)-Fenchone is a terpenoid that can be isolated from fennel oil. (±)-Fenchone has been synthesized through the following route. Supply the missing intermediates and reagents. CO2Me CO2Me...
-
The flow blockage associated with the use of an intrusive probe can be important. Determine the percentage increase in section velocity corresponding to a \(0.5 \%\) reduction in flow area due to...
-
Equipment acquired on January 8, 2011, at a cost of $420,000, has an estimated useful life of 15 years, has an estimated residual value of $30,000, and is depreciated by the straight-line method. a....
-
A light source is located 1.55m below the surface of a swimming pool and 0.545m from one edge of the pool. At what angle does the light reaching the edge of the pool leave the water? You have a piece...
-
Listed below are heights (in.) of fathers and their first sons. The data are from a journal kept by Francis Galton. (See Data Set 5 "Family Heights" in Appendix B.) Use a 0.05 significance level to...
-
The following model is representation of critic acid, the key substance in the so-called citric acid cycle by which food molecules is metabolized in the body. Only the connection between atoms is...
-
How many valence electrons does the each of the following dietary trace elements have? (a) Zinc (b) Iodine (c) Silicon (d) Iron
-
What are risks? How are objectives and threats related to risk assessment?
-
A progress report for the division of tourism promotion prepared for transactions through March 31 of the fiscal year that runs from July 1 to June 30 shows that the travel account, with a total...
-
BMX Company has one employee. FICA Social Security taxes are 6.2% of the first $137,700 paid to its employee, and FICA Medicare taxes are 1.45% of gross pay. For BMX, its FUTA taxes are 0.6% and SUTA...
-
It is 1:00 pm. Tim has waited for an hour in line to get a free Starbucks coffee. This has cost Tim $50.00 in lost wages for the hour of work he missed. He is told by the barista that it will take 30...
-
Some fringe benefits are deducted on a pre-tax basis and some fringe benefits are deducted on a post-tax basis. Is the employee better off when fringe benefits are deducted pretax basis or past tax...
-
Vikings Company pays Jim Reap gross earnings for the current two-week period of $4,500. Federal withholdings were withheld in the amount of $825.50 along with FICA (6% for Social Security and 1.5%...
-
Determine which trades are most likely to exhibit the greatest execution risk and market impact. Justify each selection. Last year, Larry Sailors left his trading position at Valley Ranch Partners, a...
-
In Problems 718, write the augmented matrix of the given system of equations. f0.01x0.03y = 0.06 [0.13x + 0.10y = 0.20
-
Prove that a. (), b. G; P,S,Nji - ( ). H - TS = G = (), aU N S,V,Nji V,T,Nji
-
Explain whether you would expect KBr or CH3Br to have the higher melting point.
-
Which of these isomers would you expect to have the higher boiling point: Explain? CH 3 CH 2 CH 2 OH or CH 3 CH 2 OCH 3
-
Which of these compounds would you expect to be more soluble in water? Explain? CHCHCHCHCOH or CH3CHCHCHCHCOH
-
A) Translate the following C into RV641 assembly. Be sure to follow the calling (Refer to slide22 of lec6_postZoom.pdf) and comment each line of your assembly. Remember that long long is 64 bits in...
-
Given a Binary Tree, find the maximum sum path from a leaf to root. Input: 1 A Input: 10 -27 4 23 Output: Explanation Following the path 3-1, results in a sum of 4, which is the maximum path sum from...
-
On December 1 , 2 0 1 5 , your company pays $ 2 4 , 0 0 0 for rent on your store that covers the next 6 months. When you paid the cash, you debited Prepaid Rent.. The adjusting journal entry on...

Study smarter with the SolutionInn App


