The following model is that of an allylic carbocation intermediate formed by Protonation of a conjugated diene
Question:
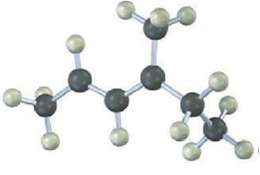
The following model is that of an allylic carbocation intermediate formed by Protonation of a conjugated diene with HBr. Show the structure of the diene and the structures of the final reactionproducts.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
HC H HC CH3 HC OH H Br C H CH3 or CH3 HC CHCH3 B...View the full answer

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The following model is that of an aldohexose: (a) Draw Fischer projections of the sugar, its enantiomer, and a diastereomer. (b) Is this a D sugar or an L sugar? Explain. (c) Draw the ? anomer of the...
-
The following model is specified:y 1 = 1 y 2 + 11 x 1 + 1 ,y 2 = 2 y 1 + 22 x 2 + 32 x 3 + 2 . All variables are measured as deviations from their means. The sample of 25 observations produces...
-
The following model is specified: y1 = 1y2 + 11x1 + 1, y2 = 2y1 + 22x2 + 32x3 + 2. All variables are measured as deviations from their means. The sample of 25 observations produces the following...
-
The following Excel output summarizes the results of an analysis of variance experiment in which the treatments were three different hybrid cars and the variable measured was the miles per gallon...
-
Suppose you work in the HR function at Nissan when it is identifying employees to work on the joint manufacturing project in Mexico. Briefly advise the company on how to prepare these employees to...
-
Baktoo Basics Ltd. is considering introducing an inventory control system that will increase inventory turnover from 10 to 15 times per year. Its annual cost of goods sold is expected to be $6...
-
A market is partially complete if for any $m$ in the market and any real function $f$, then $f(m)$ is also in the market. In addition, there are other assets that are not traded. As an example,...
-
Q1. Use the information below for J.C. Penney and Intel to answer the following questions. a. Calculate the values for (A) and (X). Revenue for INTC is more than (__________ / 10 times) greater than...
-
2.a. (1 points) What is the representation of 198.128.54.10 in binary format? 2.b. (4 points) A frame of 153 bytes is sent over a communication channel. For each of the following cases, compute the...
-
1. Consider the problem of returning change to a customer requiring the smallest number of coins. a. Show that the greedy algorithm given below returns the smallest number of coins for the coin set...
-
The following diene does not undergo Diels?Alder reactions. Explain.
-
Give IUPAC names for the followingcompounds: (b) HCHHCHCHCH3 (a) CCHH CH (e) CCHHH3 (d) CH2CH2CH3 CCH3CCH3C2
-
The adjusted trial balance for Aruns Animations is presented below. Instructions a. Prepare an income statement and statement of owners equity for the year. Mr. Arun invested $5,000 cash in the...
-
If $100 is placed in an account that earns a nominal 3 percent, compounded quarterly, what will it be worth in 5 years? Please keep two decimal places.
-
Faye has recently retired and requires an extra $2700 per year in income. She has $60,000 to invest and can invest in either low risk AA-rated bonds that pay 3% per annum or high risk B-rated junk...
-
Isabella works two part time jobs. One job is at a restaurant that pays $11 an hour and the other is delivering groceries for $17.50 an hour. Between the two jobs Isabella wants to earn at least $570...
-
Explain why we can use $ 0 . 8 6 9 8 per unit for variable OH without recalculating it ?
-
A garage service manager has authorized $535 to be spent on overtime to complete a job. If the overtime pay is $26.75 above the normal hourly pay rate, how many hours of overtime were authorized?
-
Girlie Glitter Co. is a UK-based chain of retail outlets whose products and services are marketed primarily at 313-year-old girls. The team who designed the concept took the theme of girl power as...
-
Give the structural formulas of the alkenes that, on ozonolysis, give: a. (CH3)2C=O and CH2=O b. Only (CH3CH2)2C=O c. CH3CH=O and CH3CH2CH=O d. O=CHCH2CH2CH2CH=O
-
A solution is an equimolar mixture of two volatile components A and B. Pure A has a vapor pressure of 50 torr, and pure B has a vapor pressure of 100 torr. The vapor pressure of the mixture is 85...
-
Predict the major products of the following reactions. (a) toluene + excess Cl2 (heat, pressure) (b) benzamide (PhCONH2) + Na (;oqioed NH3, CH3CH2OH) (d) o-xylene + H2 (1000 psi, 100oC, Rh catalyst)...
-
Predict the major products of treating the following compounds with hot, concentrated potassium permanganate, followed by acidification with dilute HCl. (a) isopropylbenzene (b) p-xylene (c) tetralin)
-
Propose a mechanism for the bromination of ethylbenzene shown above.
-
What are the vulnerabilities of embedded operating systems, such as, but not limited to, IoT devices, programmable logic devices, and vehicle control systems.
-
Factor the polynomial completely. 7x-17x-12
-
What are some relevant recommendations about Coca-Cola's Planning, Design, and Implementation of an Enterprise Resource Planning System that other companies can apply to their own initiatives?

Study smarter with the SolutionInn App


